
 Top of Part 4
Top of Part 4

 Last page
Last page

 Next page
Next page
  Top of Part 4
Top of Part 4
  Last page
Last page
  Next page
Next page
|
4-4: The Wave Nature of Electrons |

 As learned As learned
 in Part 1 and 2, in Part 1 and 2,
 it has been made clear it has been made clear
 that, that,
 if a substance if a substance
 is divided into is divided into
 finer and finer pieces, finer and finer pieces,
 we reach molecules we reach molecules
 and atoms, and atoms,
 and then we realize and then we realize
 that the atoms consist of that the atoms consist of
 electrons and nuclei. electrons and nuclei.
 Namely, it has been Namely, it has been
 clarified that clarified that
 matter is a collection matter is a collection
 of ultramicroscopic of ultramicroscopic
 particles. particles.
 In the classical physics In the classical physics
 up to the 19th century, up to the 19th century,
 these particles were these particles were
 considered to move considered to move
 obeying Newtonian mechanics obeying Newtonian mechanics
 and Maxwellian electromagnetism. and Maxwellian electromagnetism.
 However, this viewpoint However, this viewpoint
 has become doubtful has become doubtful
 after the proposal of
the Bohr model after the proposal of
the Bohr model
 of the atomic structure of the atomic structure
 (Bohr's quantum theory). (Bohr's quantum theory).

 On the other hand, On the other hand,
 light light
 had been considered had been considered
 to be electromagnetic waves to be electromagnetic waves
 in the classical physics. in the classical physics.
 However, after However, after
 the discovery
of light quanta (photons),
the discovery
of light quanta (photons),
 it was clarified it was clarified
 that the light has that the light has
 wave nature at one time wave nature at one time
 and and
 particle nature particle nature
 at another time. at another time.
 Namely, light has Namely, light has
 a kind of duality. a kind of duality.

 Thinking of this, Thinking of this,
 L. V. de Broglie L. V. de Broglie
 (France, 1892 - 1987) (France, 1892 - 1987)
 guessed that such substance guessed that such substance
 particles as electrons or particles as electrons or
 protons might have protons might have
 wave nature, although wave nature, although
 they had been considered they had been considered
 to have particle nature to have particle nature
 so far. so far.

|
 [De Broglie Waves] [De Broglie Waves]

 The idea of The idea of
 de Broglie waves de Broglie waves
 or or
 de Broglie matter waves de Broglie matter waves
 is that, is that,
 thinking of the fact thinking of the fact
 that light which had been that light which had been
 considered to be considered to be
 waves had the particle nature, waves had the particle nature,
 such a substance particle such a substance particle
 as electron or proton as electron or proton
 which had been considered which had been considered
 to be "particle" to be "particle"
 might have "wave nature". might have "wave nature".

 Hence, de Broglie Hence, de Broglie
 thought that, thought that,
 if the double aspect, if the double aspect,
 i.e. i.e.
 the duality the duality
 of the wave nature of the wave nature
 and the particle nature, and the particle nature,
 is valid not only is valid not only
 for light but also for light but also
 for such substance for such substance
 particles as electrons, particles as electrons,
 Einstein's relations Einstein's relations
 which connect the particle which connect the particle
 and wave aspects and wave aspects
 in light quanta in light quanta




 would be satisfied would be satisfied
 for de Broglie matter for de Broglie matter
 waves as well. waves as well.
 Therefore the relations, Therefore the relations,
 Eq. (1), Eq. (1),
 are often called are often called
 Einstein - de Broglie's relations. Einstein - de Broglie's relations.

 If we apply these relations If we apply these relations
 to the case of the Bohr model to the case of the Bohr model
 of the hydrogen atom, of the hydrogen atom,
 we can well understand we can well understand
 its plausibility its plausibility
 as follows. as follows.
 If we consider If we consider
 that the electron that the electron
 in a hydrogen atom in a hydrogen atom
 moves at constant speed moves at constant speed
 along a circular orbit along a circular orbit
 around the nucleus around the nucleus
 (proton), the
quantum condition (proton), the
quantum condition
 in Bohr's quantum theory in Bohr's quantum theory
 is written as is written as
 Eq. (3) on the preceding page. Eq. (3) on the preceding page.
 By using Einstein's relation By using Einstein's relation


 in this equation, in this equation,
 the quantum condition the quantum condition
 is written is written




 This equation means This equation means
 that the circumference that the circumference
 of the circular orbit of the circular orbit
 of the electron of the electron
 must be a integral multiple must be a integral multiple
 of the wavelength of of the wavelength of
 de Broglie wave. de Broglie wave.
 In other word, In other word,
 de Broglie wave de Broglie wave
 accompanying the motion accompanying the motion
 of the electron of the electron
 should be should be
 continuous. continuous.
 Therefore, we can easily Therefore, we can easily
 understand the quantum condition understand the quantum condition
 that determines that determines
 the stationary states the stationary states
 by considering the continuity by considering the continuity
 of de Broglie waves. of de Broglie waves.
 (See the following figure.) (See the following figure.)

|

|
 Bohr's quantum condition.
Bohr's quantum condition.
 The condition for stationary states
The condition for stationary states

 The circumference of The circumference of
 the circular orbit the circular orbit
 of the electron of the electron
 should be an integral multiple should be an integral multiple
 of the wavelength of the wavelength
 of de Broglie wave, of de Broglie wave,
 otherwise the wave otherwise the wave
 cannot be smoothly continuous. cannot be smoothly continuous.

|
 [The Laue Diagram [The Laue Diagram
 -- Wave Nature of X-Rays] -- Wave Nature of X-Rays]

 In 1914, In 1914,
 M. T. F. von Laue M. T. F. von Laue
 (Germany, 1879 - 1960) (Germany, 1879 - 1960)
 found a symmetrical pattern found a symmetrical pattern
 of spots of spots
 on a photographic plate on a photographic plate
 by a beam of X-rays by a beam of X-rays
 that had passed that had passed
 through a crystal. through a crystal.
 This is called the This is called the
 Laue diagram, Laue diagram,
 which is produced which is produced
 by interference by interference
 of beams of X-rays diffracted of beams of X-rays diffracted
 on the different atomic planes on the different atomic planes
 in the crystal. in the crystal.
 It was confirmed It was confirmed
 by this phenomena that by this phenomena that
 X-rays are electromagnetic X-rays are electromagnetic
 waves of very short wavelengths. waves of very short wavelengths.


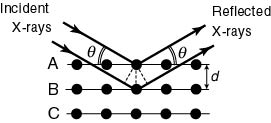

 The reason why The reason why
 the interference pattern the interference pattern
 in the Laue diagram in the Laue diagram
 appears is as follows: appears is as follows:
 As shown in the above As shown in the above
 figure, figure,
 an X-ray beam an X-ray beam
 is irradiated on a crystal is irradiated on a crystal
 consisting of the atomic consisting of the atomic
 planes A, B, ... planes A, B, ...
 As light reflects As light reflects
 on a mirror, on a mirror,
 so the X-ray beam striking so the X-ray beam striking
 the plane A the plane A
 with with
 the incident angle the incident angle
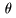
 reflects most strongly reflects most strongly
 in the same angle in the same angle
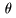 . .
 The similar happens The similar happens
 on the plane B. on the plane B.
 These X-ray beams reflected These X-ray beams reflected
 on the planes A and B on the planes A and B
 interfere with each other and interfere with each other and
 constructive interference constructive interference
 takes place when takes place when
 the difference in pathlength the difference in pathlength
 is equal to an integral is equal to an integral
 number of wavelength number of wavelength
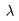 , ,
 i.e., i.e.,


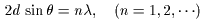
 where d is where d is
 the interplanar distance, the interplanar distance,
 and and
 is the angle is the angle
 between between
 the incident X-ray the incident X-ray
 and the crystal plane. and the crystal plane.
 This is called This is called
 Bragg's condition. Bragg's condition.
 Thus a bright spot Thus a bright spot
 will appear will appear
 in the angle satisfying in the angle satisfying
 the condition. the condition.
 When the white X-rays When the white X-rays
 which contain all wavelengths which contain all wavelengths
 are illuminated on a crystal, are illuminated on a crystal,
 various atomic planes various atomic planes
 in the crystal in the crystal
 reflect the X-rays satisfying reflect the X-rays satisfying
 Bragg's condition Bragg's condition
 selectively or exclusively, selectively or exclusively,
 and then and then
 a symmetric pattern a symmetric pattern
 of spots is produced of spots is produced
 on the photographic plate on the photographic plate
 placed behind. placed behind.
 This is the Laue diagram, This is the Laue diagram,
 an example of which an example of which
 is shown in the following is shown in the following
 picture. picture.

|
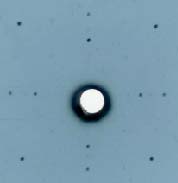
|
 An example of the Laue diagram
An example of the Laue diagram

 This is a interference This is a interference
 pattern of X-rays pattern of X-rays
 diffracted by diffracted by
 a single crystal of silicon. a single crystal of silicon.
 The small black spots The small black spots
 lined crosswise lined crosswise
 are the so-called Laue spots. are the so-called Laue spots.
 Here, the wavelength Here, the wavelength
 and the interatomic distance and the interatomic distance
 are about are about
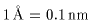
 (Courtesy of Prof. Y. Soejima, (Courtesy of Prof. Y. Soejima,
 Dept. of Physics, Dept. of Physics,
 Kyushu Univ.) Kyushu Univ.)

|
 [The Empirical Evidences [The Empirical Evidences
 of the Wave Nature of Electrons] of the Wave Nature of Electrons]

 The wavelength of The wavelength of
 de Broglie wave de Broglie wave
 associated with the electrons associated with the electrons
 accelerated by accelerated by
 an electric potential an electric potential
 of 100 V of 100 V
 is about is about
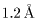
 (
(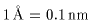 ). ).

 This is almost This is almost
 the same as that the same as that
 of ordinary X-rays. of ordinary X-rays.
 It is therefore supposed It is therefore supposed
 that, if we bombarded that, if we bombarded
 this electron beam this electron beam
 on a crystal, on a crystal,
 we would observe we would observe
 a diffraction pattern a diffraction pattern
 similar to similar to
 the Laue diagram the Laue diagram
 in the case of X-rays. in the case of X-rays.

 In 1927, In 1927,
 American physicists, American physicists,
 C. J. Davisson C. J. Davisson
 (USA, 1881 - 1958) and (USA, 1881 - 1958) and

 L. H. Germer L. H. Germer
 (USA, 1896 - 1971) (USA, 1896 - 1971)
 observed an interference observed an interference
 pattern of pattern of
 electron beams electron beams
 diffracted by a single diffracted by a single
 crystal of nickel crystal of nickel
 for the first time for the first time
 and, in the same year, and, in the same year,
 G. P. Thomson G. P. Thomson
 (UK, 1892 - 1975) (UK, 1892 - 1975)
 independently observed independently observed
 the diffraction pattern the diffraction pattern
 using a metallic multi-crystal. using a metallic multi-crystal.
 Next year, Next year,
 S. Kikuchi S. Kikuchi
 (Japan, 1902 - 74) (Japan, 1902 - 74)
 was also successful was also successful
 in a similar experiment in a similar experiment
 using a thin film of mica. using a thin film of mica.

 An example of the diffraction An example of the diffraction
 and interference and interference
 phenomena of electrons phenomena of electrons
 is shown in the following is shown in the following
 picture. picture.

|

|
A diffraction pattern of electron beams

 Electron beams Electron beams
 are diffracted are diffracted
 by a crystal by a crystal
 of manganese-nickel alloy. of manganese-nickel alloy.
 In this case, the de Broglie wavelength In this case, the de Broglie wavelength
 is shorter than is shorter than
 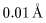
 which corresponds which corresponds
 to rather high speed to rather high speed
 electron beam. electron beam.
 (Courtesy of Prof. Y. Soejima, (Courtesy of Prof. Y. Soejima,
 Dept. of Physics, Dept. of Physics,
 Kyushu Univ.) Kyushu Univ.)

|
 Looking at these results, Looking at these results,
 people could no longer people could no longer
 deny that deny that
 electrons possess electrons possess
 the double nature the double nature
 (dual property), (dual property),
 i.e. the particle nature i.e. the particle nature
 and the wave nature. and the wave nature.

|
 Top
Top
|
|

 Go back to
the top page of Part 3. Go back to
the top page of Part 3.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|