
 Top of Part 3
Top of Part 3

 Last page
Last page

 Next page
Next page
  Top of Part 3
Top of Part 3
  Last page
Last page
  Next page
Next page
|
3-2: The Structure of Hydrogen Atom |
 As mentioned before, As mentioned before,
 it is considered that it is considered that
 there is a heavy there is a heavy
 proton at the center of proton at the center of
 a hydrogen atom a hydrogen atom
 and a light electron and a light electron
 revolves around it. revolves around it.
 The proton may be thought The proton may be thought
 to be approximately to be approximately
 at rest at the origin at rest at the origin
 of the coordinate of the coordinate
 (the center of the (the center of the
 hydrogen atom) hydrogen atom)
 because proton is because proton is
 about 1800 times heavier about 1800 times heavier
 than electron. than electron.

 The Coulomb attractive force The Coulomb attractive force
 works between the proton works between the proton
 and the electron. and the electron.
 Its potential is written Its potential is written


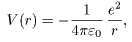
 where r where r
 is the distance between is the distance between
 the proton and the electron. the proton and the electron.
 The Schroedinger equation The Schroedinger equation
 describing the motion describing the motion
 of the electron is of the electron is
 given by given by


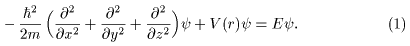
 Needless to say, Needless to say,
 the wave function the wave function
 is a function is a function
 of of
 x , x ,
 y y
 and and
 z . z .

|
 [The Eigenstates of Hydrogen Atom] [The Eigenstates of Hydrogen Atom]
 In the present case, In the present case,
 the polar coordinate the polar coordinate

 shown in the following shown in the following
 Fig. (A) Fig. (A)
 is more convenient than is more convenient than
 the Cartesian coordinate the Cartesian coordinate
 . .



 We solve We solve
 the Schroedinger equation the Schroedinger equation
 (1) represented (1) represented
 in the polar coordinate in the polar coordinate

 setting the boundary condition setting the boundary condition
 that the wave function that the wave function
 should be smoothly continuous should be smoothly continuous
 at every point of at every point of
 the coordinate space the coordinate space
 and should converge to 0 and should converge to 0
 at the infinitely long distance at the infinitely long distance
 . .
 Then we have Then we have
 a set of discrete a set of discrete
 energy eigenvalues energy eigenvalues
 and the corresponding eigenstates. and the corresponding eigenstates.
 The details of The details of
 the method to solve it is the method to solve it is
 omitted here. omitted here.
 If you want to study them, If you want to study them,
 please refer to some please refer to some
 other textbooks other textbooks
 of quantum mechanics. of quantum mechanics.

 The wave functions The wave functions
 of the eigenstates of the eigenstates
 is expressed as is expressed as


 Here the part Here the part

 is called the is called the
 radial wave function radial wave function
 which is specified which is specified
 by a set of integers, by a set of integers,
 n n
 and and
 l . l .
 Such numbers (integers) as these Such numbers (integers) as these
 n n
 and and
 l l
 are sometimes called are sometimes called
 quantum numbers, quantum numbers,
 which characterize which characterize
 the eigenstates. the eigenstates.

 In the present case, they are In the present case, they are



 The part The part

 denotes the angular denotes the angular
 wave function wave function
 which is specified which is specified
 by a set of quantum by a set of quantum
 numbers (integers), numbers (integers),
 l l
 and and
 m , and m , and
 they are given by they are given by



 The angular wave function The angular wave function

 describes the revolving state describes the revolving state
 of the electron around of the electron around
 the coordinate origin (proton). the coordinate origin (proton).
 Namely, Namely,
 the quantum number the quantum number
 l l
 expresses the speed expresses the speed
 of the revolution of of the revolution of
 the electron, the electron,
 i.e. the magnitude of i.e. the magnitude of
 the angular momentum the angular momentum
 of the electron, and of the electron, and
 m m
 represents represents
 the orientation (direction) the orientation (direction)
 of angular momentum vector. of angular momentum vector.
 The fact that these quantum numbers The fact that these quantum numbers
 l l
 and and
 m m
 are integers are integers
 means that means that
 both the magnitude both the magnitude
 and and
 the orientation the orientation
 of the angular momentum of the angular momentum
 are step-like and discrete. are step-like and discrete.

 This result implies that This result implies that
 not only energy but also not only energy but also
 angular momentum angular momentum
 and its orientation and its orientation
 are quantized are quantized
 in quantum mechanics. in quantum mechanics.
 This was confirmed This was confirmed
 by the Stern-Gerlach experiment by the Stern-Gerlach experiment
 (1922). (1922).
 Needless to say, Needless to say,
 this also originates this also originates
 from the particle-wave from the particle-wave
 duality of electrons. duality of electrons.
 And this can never understood And this can never understood
 by the classical theory. by the classical theory.

|
 [The Energy Eigenvalues of Hydrogen Atom]
[The Energy Eigenvalues of Hydrogen Atom]
 The energy eigenvalues The energy eigenvalues
 of hydrogen atom of hydrogen atom
 are determined are determined
 only by the quantum number only by the quantum number
 n n
 and they are and they are
 expressed as expressed as


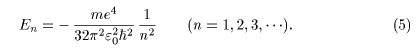
 The state with n The state with n
 = 1 = 1
 is the lowest energy state is the lowest energy state
 (the ground state) (the ground state)
 and those with and those with
 n n
 = 2, 3, ... = 2, 3, ...
 are the excited states. are the excited states.
 Thus the ground-state energy Thus the ground-state energy
 is written is written


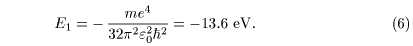
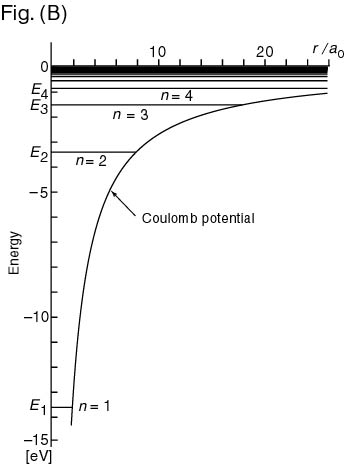
 The energy eigenvalues The energy eigenvalues
 (energy levels) (energy levels)
 are shown in the following are shown in the following
 Fig. (B), Fig. (B),
 in which in which
 the heights of the the heights of the
 horizontal lines (levels) horizontal lines (levels)
 show the energy show the energy
 eigenvalues obtained eigenvalues obtained
 by solving the Schroedinger equation by solving the Schroedinger equation
 (1). (1).

|
|
 Fig. (B): The energy eigenvalues Fig. (B): The energy eigenvalues
 of hydrogen atom of hydrogen atom

 The abscissa denotes The abscissa denotes
 the position coordinate the position coordinate
 of the electron of the electron
 (the distance between (the distance between
 the proton and electron), the proton and electron),
 r , r ,
 in units of the in units of the
 Bohr radius Bohr radius
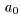 , ,
 where where


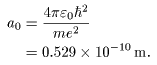
|
 [The Probability [The Probability
 that Electron will be Found] that Electron will be Found]

 It is very interesting It is very interesting
 to see how much to see how much
 the probability the probability
 for a electron to be found for a electron to be found
 at a point in the space is. at a point in the space is.
 The probability density The probability density


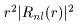
 at a position of the distance at a position of the distance
 r r
 from the center from the center
 is shown in is shown in
 Fig. (C). Fig. (C).
 Integrating this probability density Integrating this probability density
 over the whole space, we have over the whole space, we have


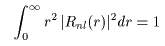
 As shown in this integration, As shown in this integration,
 the present wave functions the present wave functions
 are so normalized are so normalized
 that the total that the total
 probability would be 1 probability would be 1
 (= 100 %). (= 100 %).


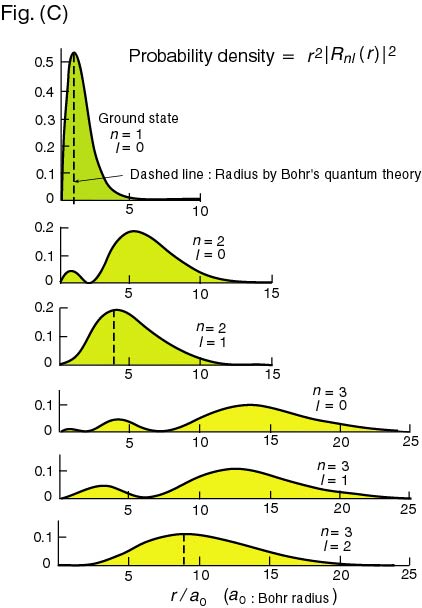
 The above Fig. (C) shows The above Fig. (C) shows
 the probability distribution the probability distribution
 of electron in hydrogen atom. of electron in hydrogen atom.
 The first figure The first figure
 shows the ground state, shows the ground state,
 in which in which
 almost all probability almost all probability
 concentrated concentrated
 at at


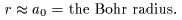
 We can therefore consider We can therefore consider
 that the true radius that the true radius
 of a hydrogen atom is of a hydrogen atom is
 almost equal to almost equal to
 the Bohr radius the Bohr radius
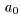 . .
 However, in the excited states, However, in the excited states,
 the probability is the probability is
 widely extended widely extended
 at the distant area, at the distant area,
 so that the excited so that the excited
 hydrogen atom hydrogen atom
 would be rather swollen. would be rather swollen.

|
 [Conclusion of This Page] [Conclusion of This Page]
 We have studied We have studied
 how the solutions how the solutions
 of the Schroedinger equation of the Schroedinger equation
 for the case of hydrogen atom are. for the case of hydrogen atom are.
 All of Bohr's quantum theory All of Bohr's quantum theory
 are completely included are completely included
 in the solutions. in the solutions.
 This means that This means that
 all the structure all the structure
 of hydrogen atom of hydrogen atom
 can perfectly be derived can perfectly be derived
 just from the single equation, just from the single equation,
 the Schroedinger equation. the Schroedinger equation.
 Thus we can understand Thus we can understand
 how splendid and how fundamental how splendid and how fundamental
 the theory of Quantum Mechanics is. the theory of Quantum Mechanics is.

|
 Top
Top
|
|

 Go back to
the top page of Part 3. Go back to
the top page of Part 3.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|