
 Top of Part 1
Top of Part 1

 Last page
Last page

 Next page
Next page
  Top of Part 1
Top of Part 1
  Last page
Last page
  Next page
Next page
|
1-3: The Discovery of the Neutron |
 [Problem of Isotopes] [Problem of Isotopes]

 J.J. Thomson J.J. Thomson
 (UK, 1856-1940) discovered (UK, 1856-1940) discovered
 that the natural neon gas that the natural neon gas
 is a mixture of two kinds of is a mixture of two kinds of
 elements which have different elements which have different
 atomic weights (1912). atomic weights (1912).
 Afterwards, F.W. Aston Afterwards, F.W. Aston
 (UK, 1877-1945) carried out (UK, 1877-1945) carried out
 precise mass-spectroscopic precise mass-spectroscopic
 analyses and analyses and
 he confirmed that he confirmed that
 many various elements many various elements
 are mixtures of two or more are mixtures of two or more
 kinds of isotopes kinds of isotopes
 which differ in mass which differ in mass
 (atomic weight). (atomic weight).
 Namely, Namely,
 there exist some elements there exist some elements
 (or atoms) which have the same atomic (or atoms) which have the same atomic
 number and, number and,
 therefore, essentially therefore, essentially
 the same chemical properties the same chemical properties
 but different atomic masses. but different atomic masses.
 This means that there exist This means that there exist
 different nuclei having the same different nuclei having the same
 electric charge but electric charge but
 different masses. different masses.
 These are also called isotopes. These are also called isotopes.

 What is What is
 a reasonable explanation a reasonable explanation
 of the existence of isotopes ? of the existence of isotopes ?

|
 [Does the Nucleus Consist of [Does the Nucleus Consist of
 Protons and Electrons?] Protons and Electrons?]

 It is known well that It is known well that
 some kinds of nucleus decay some kinds of nucleus decay
 by emitting electrons. by emitting electrons.
 This is called the beta decay. This is called the beta decay.
 If we consider that the atomic nucleus If we consider that the atomic nucleus
 consists of protons and consists of protons and
 electrons, then the beta decay could electrons, then the beta decay could
 easily be understood. easily be understood.
 However, the idea that protons and However, the idea that protons and
 electrons are basic constituents electrons are basic constituents
 of a nucleus has the following of a nucleus has the following
 serious difficulties. serious difficulties.

|
 (1) Problem on the spin (1) Problem on the spin
 and the statistics and the statistics

 As for the details of As for the details of
 the spin and the statistics, the spin and the statistics,
 please refer to some other please refer to some other
 advanced books concerning advanced books concerning
 "Quantum Mechanics". "Quantum Mechanics".

 Here, let us give only Here, let us give only
 a rough outline of the discussion a rough outline of the discussion
 about the present problem. about the present problem.
 (See the page of (See the page of
 "Microscopic World -2-: "Microscopic World -2-:
 [The Pauli Principle, Spin]" ).
[The Pauli Principle, Spin]" ).

 For simplicity of For simplicity of
 our explanation, let us take our explanation, let us take
 as an example as an example
 the case of nitrogen-14 nucleus
( the case of nitrogen-14 nucleus
(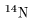 ) )
 whose atomic number is 7 whose atomic number is 7
 and atomic weight is 14. and atomic weight is 14.

 A single proton and A single proton and
 a single electron a single electron
 are both are both
 fermion. fermion.
 A composit system of even number A composit system of even number
 of fermions is a boson of fermions is a boson
 as a whole. as a whole.
 And it has the property of And it has the property of
 Bose-Einstein statistics. Bose-Einstein statistics.
 Contrarily, a composit system Contrarily, a composit system
 of odd number of fermions of odd number of fermions
 behaves as a fermion behaves as a fermion
 as a whole, as a whole,
 and it has the property of and it has the property of
 Fermi-Dirac statistics. Fermi-Dirac statistics.

 If we consider that If we consider that
 nitrogen-14 nucleus
( nitrogen-14 nucleus
(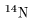 ) )
 consists of 14 protons consists of 14 protons
 and 7 electrons, and 7 electrons,
 the total particle number is 21 the total particle number is 21
 that is an odd number. that is an odd number.
 In contrast to this, In contrast to this,
 the experimental result the experimental result
 of the spectroscopy of the spectroscopy
 shows that the nitrogen-14 nucleus shows that the nitrogen-14 nucleus
 must be a boson as a whole. must be a boson as a whole.
 This is the first problem. This is the first problem.

 The second problem is as follows: The second problem is as follows:
 Each of the proton and the electron Each of the proton and the electron
 has spin has spin
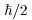 . .
 While the total spin of While the total spin of
 a system of even number of protons a system of even number of protons
 (or electrons) (or electrons)
 is an integral multiple of is an integral multiple of
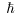 , ,
 that of odd-proton (or odd-electron) system that of odd-proton (or odd-electron) system
 must be an odd-integral multiple of must be an odd-integral multiple of

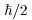 . .
 However, the experimental data show However, the experimental data show
 that that
 the spin of the nitrogen-14 nucleus the spin of the nitrogen-14 nucleus
 is 0 (= even). is 0 (= even).
 This means that total particle number This means that total particle number
 of the nitrogen-14 nucleus of the nitrogen-14 nucleus
 must be even. must be even.
 This is also an important problem. This is also an important problem.

 (2) Problem concerning (2) Problem concerning
 the uncertainty relation the uncertainty relation

 If we consider that If we consider that
 the atomic nucleus consists of the atomic nucleus consists of
 protons and electrons, protons and electrons,
 an alpha particle an alpha particle
 (= helium nucleus) (= helium nucleus)
 should be made of 4 protons should be made of 4 protons
 and 2 electrons. and 2 electrons.
 The size (diameter) of the The size (diameter) of the
 alpha particle is about alpha particle is about

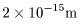 . .

 If an electron is confined If an electron is confined
 within this size of space, within this size of space,
 the uncertainty of the position the uncertainty of the position
 of the electron should be of the electron should be
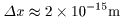 . .

 Using the uncertainty Using the uncertainty
 relation in "Quantum Mechanics", relation in "Quantum Mechanics",

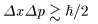 , ,
 we can estimate the uncertainty we can estimate the uncertainty
 of the momentum of the electron, of the momentum of the electron,
 and then the and then the
 uncertainty of the speed of uncertainty of the speed of
 the electron will be the electron will be

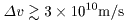 . .
 This value largely exceeds This value largely exceeds
 the light speed. the light speed.
 This is quite unreasonable This is quite unreasonable
 on the basis of on the basis of
 the Special Theory of Relativity. the Special Theory of Relativity.
 Namely, it is completely impossible Namely, it is completely impossible
 to confine such a light particle to confine such a light particle
 as electron within as electron within
 a narrow space like a nucleus. a narrow space like a nucleus.
 This is another serious problem. This is another serious problem.

|


|
 [Rutherford's Prediction] [Rutherford's Prediction]

 As discussed above, As discussed above,
 the problems of the idea the problems of the idea
 that the nucleus consists of that the nucleus consists of
 protons and electrons protons and electrons
 could at all once be solved could at all once be solved
 if we assume that if we assume that
 there exists an electrically there exists an electrically
 neutral particle with neutral particle with
 almost the same mass as a proton; almost the same mass as a proton;
 so to say a "neutral proton". so to say a "neutral proton".
 If it is so, the existence of If it is so, the existence of
 isotopes could easily be understood. isotopes could easily be understood.

 This was Rutherford's This was Rutherford's
 prediction and he sometimes prediction and he sometimes
 talked this idea to his talked this idea to his
 students in his lecture. students in his lecture.
 One of his top students One of his top students
 influenced strongly by him, influenced strongly by him,
 J. Chadwick J. Chadwick
 (UK, 1891-1974), (UK, 1891-1974),
 made an effort to find out made an effort to find out
 that "neutral proton" that "neutral proton"
 and finally got the honor and finally got the honor
 of discovering that particle; of discovering that particle;
 Chadwick named it "neutron". Chadwick named it "neutron".

|
 [History of the Discovery [History of the Discovery
 of the Neutron] of the Neutron]

 The discovery of the neutron The discovery of the neutron
 was based on the results of efforts was based on the results of efforts
 by some research groups by some research groups
 for several years. for several years.

|
 (1) Experiment of Bothe and Becker (1) Experiment of Bothe and Becker

 W. Bothe (Germany, 1891-1975) W. Bothe (Germany, 1891-1975)
 and his student H. Becker and his student H. Becker
 bombarded beryllium with bombarded beryllium with
 strong alpha-rays from a polonium source strong alpha-rays from a polonium source
 and found a high-energy radiation and found a high-energy radiation
 of strong penetrability (1928). of strong penetrability (1928).
 They observed similar radiations They observed similar radiations
 not only from beryllium, not only from beryllium,
 but also from lithium and boron. but also from lithium and boron.
 At first, these radiations At first, these radiations
 of strong penetrability of strong penetrability
 were considered to be high-energy were considered to be high-energy
 gamma rays. gamma rays.

 (2) Experiment of I. Curie and Joliot (2) Experiment of I. Curie and Joliot

 Around 1931, Around 1931,
 I. Curie (France, 1897-1956) I. Curie (France, 1897-1956)
 and F. Joliot and F. Joliot
 (France, 1900-58) (France, 1900-58)
 reported that the above reported that the above
 high-energy radiation found high-energy radiation found
 by Bothe and Becker by Bothe and Becker
 was able to eject was able to eject
 protons from a paraffin layer. protons from a paraffin layer.
 I. and F. Joliot-Curie I. and F. Joliot-Curie
 thought that this phenomenon thought that this phenomenon
 is caused by the Compton effect is caused by the Compton effect
 due to high-energy gamma-rays due to high-energy gamma-rays
 emitted from the polonium-beryllium emitted from the polonium-beryllium
 source. source.
 Namely, they thought that Namely, they thought that
 a gamma particle (photon) a gamma particle (photon)
 of Bothe-Becker's high-energy of Bothe-Becker's high-energy
 radiation hits a proton radiation hits a proton
 in the paraffin layer in the paraffin layer
 to be scattered to be scattered
 and the observed proton and the observed proton
 is nothing but the recoil proton is nothing but the recoil proton
 from this Compton effect. from this Compton effect.
 (As for the Compton effect, refer to (As for the Compton effect, refer to
 the page of the page of
 "Microscopic World -1-: "Microscopic World -1-:
 3-7:The Compton Effect").
3-7:The Compton Effect").

 (3) The Discovery of the Neutron (3) The Discovery of the Neutron
 by Chadwick by Chadwick

 Chadwick was much interested Chadwick was much interested
 in the report of Joliot-Curie. in the report of Joliot-Curie.
 He repeated the experiments, He repeated the experiments,
 using the polonium-beryllium using the polonium-beryllium
 (Po-Be) source. (Po-Be) source.

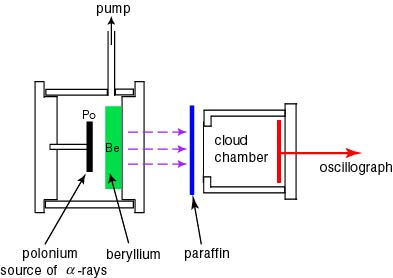
 The schematic drawing The schematic drawing
 of his experimental apparatus of his experimental apparatus
 is shown in a figure below. is shown in a figure below.
 The high-energy radiation The high-energy radiation
 emitted from the (Po-Be) source emitted from the (Po-Be) source
 on the left-hand side on the left-hand side
 is led to the is led to the
 cloud chamber,
cloud chamber,
 in front of which in front of which
 a paraffin layer is placed. a paraffin layer is placed.
 The radiation is scattered The radiation is scattered
 by a proton in the parafin by a proton in the parafin
 and a photograph of the recoil proton and a photograph of the recoil proton
 is taken in the cloud chamber is taken in the cloud chamber
 on the right-hand side. on the right-hand side.

 Chadwick collided the Chadwick collided the
 radiation radiation
 emerging from the (Po-Be) source emerging from the (Po-Be) source
 not only with proton (paraffin), not only with proton (paraffin),
 but also with helium and nitrogen. but also with helium and nitrogen.
 Comparing the results of these Comparing the results of these
 experiments with each other, experiments with each other,
 Chadwick concluded that Chadwick concluded that
 this mysterious radiation from the (Po-Be) this mysterious radiation from the (Po-Be)
 source cannot be interpreted source cannot be interpreted
 by assuming it to be a gamma ray. by assuming it to be a gamma ray.
 He finally concluded that He finally concluded that
 all were able to be understood all were able to be understood
 without any contradiction without any contradiction
 by assuming that by assuming that
 the mysterious radiation is the mysterious radiation is
 electrically neutral particles electrically neutral particles
 with almost the same with almost the same
 mass as a proton. mass as a proton.
 This is the confirmation This is the confirmation
 of the existence of of the existence of
 the "neutral proton" predicted the "neutral proton" predicted
 by Rutherford. by Rutherford.
 Chadwick named this particle Chadwick named this particle
 "neutron" (1932). "neutron" (1932).

|
|
 [Experimental Apparatus of Chadwick] [Experimental Apparatus of Chadwick]

 This is a schematic drawing This is a schematic drawing
 of the experimental apparatus of the experimental apparatus
 of Chadwick. of Chadwick.
 High-energy alpha rays High-energy alpha rays
 from polonium from polonium
 on the left-hand side on the left-hand side
 collide the beryllium target collide the beryllium target
 and a radiation with and a radiation with
 strong penetrability strong penetrability
 (purple arrows (purple arrows
 = neutrons in fact) = neutrons in fact)
 comes out to hit the proton comes out to hit the proton
 in the paraffin layer. in the paraffin layer.
 The knocked proton is The knocked proton is
 observed in the cloud chamber observed in the cloud chamber
 behind. behind.

|
 Top
Top
|
|

 Go back to
the top page of Part 1. Go back to
the top page of Part 1.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|