
 Top of Part 2
Top of Part 2

 Last page
Last page

 Next page
Next page
  Top of Part 2
Top of Part 2
  Last page
Last page
  Next page
Next page
|
2-4: The Liquid Drop Model |
 Needless to say, Needless to say,
 a nucleus is a quantum mechanical a nucleus is a quantum mechanical
 many-body system many-body system
 consisting of many nucleons. consisting of many nucleons.
 We can not directly watch it We can not directly watch it
 with our eyes. with our eyes.
 In such case, In such case,
 how can we see the structure how can we see the structure
 of a nucleus? of a nucleus?
 Of course, the fundamental Of course, the fundamental
 theory to govern the nuclear structure theory to govern the nuclear structure
 is Quantum Mechnics, is Quantum Mechnics,
 and the basic and the basic
 equation to describe it equation to describe it
 must be the must be the
 Schroedinger equation. Schroedinger equation.
 It is however very difficult and It is however very difficult and
 almost impossible to almost impossible to
 precisely solve precisely solve
 the many-body Schroedinger the many-body Schroedinger
 equation for such a equation for such a
 complicated system as nuclei. complicated system as nuclei.

 For such a complicated For such a complicated
 system, we sometimes assume system, we sometimes assume
 a simple model a simple model
 which is very easy which is very easy
 to treat and understand. to treat and understand.
 If this model can reproduce If this model can reproduce
 a lot of experimental data a lot of experimental data
 on nuclei in a unified way, on nuclei in a unified way,
 then we could then we could
 understand the structure of understand the structure of
 the nuclei. the nuclei.

 Here, let us discuss Here, let us discuss
 the liquid drop model, the liquid drop model,
 which was the oldest one which was the oldest one
 introduced for the nuclear introduced for the nuclear
 structure. structure.

|
 [The Liquid Drop Model] [The Liquid Drop Model]

 Just after the discovery Just after the discovery
 of the neutron by Chadwick, of the neutron by Chadwick,
 Ivanenko and Heisenberg proposed Ivanenko and Heisenberg proposed
 an idea that nuclei an idea that nuclei
 are composed of protons are composed of protons
 and neutrons (1932). and neutrons (1932).

 Then it becomes the next question Then it becomes the next question
 what structure do nuclei have. what structure do nuclei have.

 C. Fv. Weizsaecker C. Fv. Weizsaecker
 (Germany, 1912- ) imaged that (Germany, 1912- ) imaged that
 the nucleus might be something the nucleus might be something
 like a raindrop. like a raindrop.
 This is called a This is called a
 liquid drop model. liquid drop model.

|
 [Weizsaecker-Bethe's Mass Formula] [Weizsaecker-Bethe's Mass Formula]

 On the basis of the On the basis of the
 liquid drop model, liquid drop model,
 Weizsaecker and Weizsaecker and
 H. A. Bethe H. A. Bethe
 (Germany, USA, 1906-2005) (Germany, USA, 1906-2005)
 proposed a simple formula proposed a simple formula
 for the nuclear binding energy. for the nuclear binding energy.

 Let the proton and neutron Let the proton and neutron
 masses be masses be
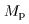
 and and
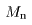 , respectively. , respectively.
 And let the mass of nucleus And let the mass of nucleus
 of the proton number Z of the proton number Z
 and the neutron number N and the neutron number N
 be be
 M (Z, N M (Z, N
 ). ).
 As learned on the
the last page, As learned on the
the last page,
 the binding energy the binding energy
 of a nucleus, of a nucleus,
 B (Z, N ), B (Z, N ),
 is given by is given by

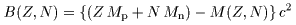

 Weizsaecker and Bethe Weizsaecker and Bethe
 found that the above found that the above
 binding energy binding energy
 may approximately be obtained by may approximately be obtained by
 a formula, a formula,
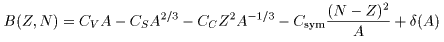

 This is the famous This is the famous
 Weizsaecker-Bethe's Mass Formula, Weizsaecker-Bethe's Mass Formula,
 which is sometimes called the which is sometimes called the
 semiempirical mass formula. semiempirical mass formula.

 Meaning of each term in the above Meaning of each term in the above
 mass formula mass formula
 can be understood can be understood
 according to the idea of according to the idea of
 the liquid drop model. the liquid drop model.

 The first term The first term
 is proportional to the nuclear volume is proportional to the nuclear volume
 and called the volume energy, and called the volume energy,
 which is the main part of which is the main part of
 the nuclear binding energy. the nuclear binding energy.

 The second term is proportional The second term is proportional
 to the area of the nuclear surface, to the area of the nuclear surface,
 and it is considered as the and it is considered as the
 surface energy surface energy
 due to the surface tension. due to the surface tension.

 The third term is The third term is
 the Coulomb energy the Coulomb energy
 which is the energy loss which is the energy loss
 due to the Coulomb repulsive forces due to the Coulomb repulsive forces
 between protons. between protons.
 Among the nuclei with Among the nuclei with
 the same mass number A the same mass number A
 , the energy loss due to the , the energy loss due to the
 Coulomb forces becomes larger Coulomb forces becomes larger
 in nuclei in nuclei
 with larger proton number Z. with larger proton number Z.
 Because of this Coulomb energy, Because of this Coulomb energy,
 nuclei with nuclei with
 larger N compared to larger N compared to
 Z Z
 have less energy loss, have less energy loss,
 so that, in the larger so that, in the larger
 mass number region, mass number region,
 the neutron number N the neutron number N
 is, in general, is, in general,
 greater than the proton number greater than the proton number
 Z Z
 . .

 The next term, the fourth term, The next term, the fourth term,
 shows an opposite effect; shows an opposite effect;
 namely, there is a property namely, there is a property
 that, the less the difference that, the less the difference
 between the proton number between the proton number
 Z Z
 and the neutron number and the neutron number
 N N
 is, is,
 the greater the energy gain becomes. the greater the energy gain becomes.
 This effect is represented by the fourth term, This effect is represented by the fourth term,
 which is called the which is called the
 symmetry energy. symmetry energy.
 This effect comes from This effect comes from
 the symmetry property the symmetry property
 in the nuclear force. in the nuclear force.

 In the nucleus, In the nucleus,
 a pair of equivalent nucleons a pair of equivalent nucleons
 (proton-proton or neutron-neutron) (proton-proton or neutron-neutron)
 couples with each other couples with each other
 more strongly than more strongly than
 a pair of non-equivalent nucleons a pair of non-equivalent nucleons
 (proton-neutron). (proton-neutron).
 This is also due to the This is also due to the
 property of the nuclear force. property of the nuclear force.
 The last term, the fifth term, The last term, the fifth term,
 represents this effect, represents this effect,
 which is called the which is called the
 even-odd mass difference. even-odd mass difference.

 The more detailed discussions The more detailed discussions
 about the last two terms, about the last two terms,
 the symmetry energy and the the symmetry energy and the
 even-odd mass difference, even-odd mass difference,
 are omitted here, because they are omitted here, because they
 would be very specialized would be very specialized
 and complicated. and complicated.

 The parameters in the The parameters in the
 above Weizsaecker-Bethe mass formula above Weizsaecker-Bethe mass formula
 should be adjusted to the experimental should be adjusted to the experimental
 nuclear masses. nuclear masses.
 The results are The results are

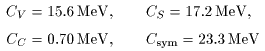
 and and

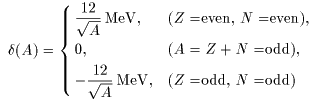
|
 [Comparison with Experimetal Data] [Comparison with Experimetal Data]

 In order to confirm In order to confirm
 how well the Weizsaecker-Bethe how well the Weizsaecker-Bethe
 mass formula can reproduce mass formula can reproduce
 the experimental data of the experimental data of
 nuclear binding energies, nuclear binding energies,
 the comparison with experiment the comparison with experiment
 is shown is shown
 in the following figure, in the following figure,
 in which the in which the
 red curve red curve
 shows the values of the shows the values of the
 binding energy per nucleon binding energy per nucleon
 obtained by obtained by
 the Weizsaecker-Bethe formula, the Weizsaecker-Bethe formula,
 and the black points indicate and the black points indicate
 the experimental data, the experimental data,
 which are equivalent to which are equivalent to
 those in
the figure those in
the figure
 at the bottom at the bottom
 of the preceding page. of the preceding page.
 You should be careful You should be careful
 because the scales of ordinates in these because the scales of ordinates in these
 two figures are somewhat varied two figures are somewhat varied
 for the sake of convenience. for the sake of convenience.

|
 As seen in the above figure, As seen in the above figure,
 the Weizsaecker-Bethe mass formula the Weizsaecker-Bethe mass formula
 can reproduce well the can reproduce well the
 experimental data experimental data
 for a wide range of nuclei. for a wide range of nuclei.
 We can therefore conclude that We can therefore conclude that
 the liquid drop model the liquid drop model
 is enough valid in nuclei. is enough valid in nuclei.

|
 Top
Top
|
|

 Go back to
the top page of Part 2. Go back to
the top page of Part 2.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|