
 Top of Part 2
Top of Part 2

 Last page
Last page

 Next page
Next page
  Top of Part 2
Top of Part 2
  Last page
Last page
  Next page
Next page
|
2-6: The Nuclear Shell Model |
 As learned on the preceding page, As learned on the preceding page,
 the nuclei whose the nuclei whose
 proton number Z proton number Z
 or or
 neutron number N neutron number N
 is one of the is one of the
 magic numbers magic numbers

 2, 8, 20, 28, 50, 82, 126
2, 8, 20, 28, 50, 82, 126
 are especially stable are especially stable
 and and
 they correspond to they correspond to
 the noble gases the noble gases
 in the atomic world. in the atomic world.
 The periodic law The periodic law
 and the periodic table and the periodic table
 are valid in the Atomic World. are valid in the Atomic World.
 Similarly, Similarly,
 the idea of the periodic table the idea of the periodic table
 in the nuclear world in the nuclear world
 was also proposed was also proposed
 in a rather early time. in a rather early time.

|
 [Why the Periodic Law] [Why the Periodic Law]

 As discussed on the page As discussed on the page
 in the present seminar, in the present seminar,
 "Microscopic World -2-:
3-3: The Periodic Law of Elements", "Microscopic World -2-:
3-3: The Periodic Law of Elements",
 the periodic law of elements the periodic law of elements
 could be interpreted could be interpreted
 with the shell structure with the shell structure
 of the atoms. of the atoms.

 Let us review the Let us review the
 atomic shell structure atomic shell structure
 again. again.

 The fundamental properties The fundamental properties
 of atoms are determined by of atoms are determined by
 the structure of electron distributions the structure of electron distributions
 surrounding the nucleus surrounding the nucleus
 at the center of atom. at the center of atom.
 Namely, they are determined by Namely, they are determined by
 the electron number the electron number
 and their arrangements and their arrangements
 (i.e. configurations). (i.e. configurations).

 The electrons in an atom The electrons in an atom
 make a layer structure make a layer structure
 starting from the central nucleus starting from the central nucleus
 and going outward. and going outward.
 Each of these layers is called Each of these layers is called
 a shell, and a shell, and
 this is none other than this is none other than
 the shell structure. the shell structure.

 An electron in atoms An electron in atoms
 is bound by the attractive is bound by the attractive
 average potential average potential
 whose main part is whose main part is
 the Coulomb force between the Coulomb force between
 the positive charge of the nucleus the positive charge of the nucleus
 and the negative charge of the electron. and the negative charge of the electron.
 The electrons bound in the atom The electrons bound in the atom
 by the average potential by the average potential
 are staying in are staying in
 eigenstates with eigenstates with
 discrete energy eigenvalues. discrete energy eigenvalues.
 These eigenstates form These eigenstates form
 the shell structure the shell structure
 of which mechanism was explained of which mechanism was explained
 in detail in detail
 on the page, on the page,
 "Microscopic World -2-:
3-3: The Periodic Law of Elements". "Microscopic World -2-:
3-3: The Periodic Law of Elements".
 In this shell structure, In this shell structure,
 each shell is occupied by each shell is occupied by
 a certain number of electrons a certain number of electrons
 according to the according to the
 Pauli principle. Pauli principle.
 When one of the shells is When one of the shells is
 filled up, a noble gas filled up, a noble gas
 is formed. is formed.
 This is the reason why the This is the reason why the
 periodic law appears. periodic law appears.

|
 [The Nuclear Shell Model] [The Nuclear Shell Model]

 The idea of the nuclear The idea of the nuclear
 shell model shell model
 had been proposed had been proposed
 at a rather early time at a rather early time
 on the analogy of on the analogy of
 the atomic shell structure. the atomic shell structure.
 However, there was some doubt However, there was some doubt
 on this idea because on this idea because
 there were differences there were differences
 between the cases between the cases
 of atoms and nuclei. of atoms and nuclei.
 The differences are as follows: The differences are as follows:

|
 (1) (1) |
 In the case of an atom, In the case of an atom,
 there is a heavy nucleus there is a heavy nucleus
 at the central position of the system, at the central position of the system,
 and it attracts and it attracts
 light electrons around light electrons around
 by the Coulomb interaction. by the Coulomb interaction.
 In contrast to this, In contrast to this,
 there is nothing special there is nothing special
 at the nuclear center. at the nuclear center.

|
 (2) (2) |
 In the case of a nucleus, In the case of a nucleus,
 it is clear that the whole system it is clear that the whole system
 can be described can be described
 in the image of in the image of
 a "raindrop". a "raindrop".
 Namely, the liquid drop model Namely, the liquid drop model
 is valid for nuclei. is valid for nuclei.
 However, such an image is not However, such an image is not
 valid for atoms. valid for atoms.

|
 Moreover, the biggest problem Moreover, the biggest problem
 is the difference is the difference
 in the magic numbers in the magic numbers
 of both the systems, of both the systems,
 atoms and nuclei. atoms and nuclei.
 Although Although
 the magic numbers in atoms are the magic numbers in atoms are

 Z Z
 = =
 2, 10, 18, 36, 54, 86,
2, 10, 18, 36, 54, 86,

 the magic numbers in nuclei are the magic numbers in nuclei are

 Z Z
 or N or N
 = =
 2, 8, 20, 28, 50, 82, 126.
2, 8, 20, 28, 50, 82, 126.

 There is a big difference There is a big difference
 between these two kinds of systems, between these two kinds of systems,
 atoms and nuclei. atoms and nuclei.
 Even if the shell structure Even if the shell structure
 is held in both the systems, is held in both the systems,
 the average potentials the average potentials
 are presumed to be are presumed to be
 considerably different. considerably different.

 In the case of atoms, In the case of atoms,
 if we assume an average potential if we assume an average potential
 consisting of the Coulomb consisting of the Coulomb
 attractive potential attractive potential
 acting from the +Ze acting from the +Ze
 charge of the nucleus charge of the nucleus
 (and the averaged Coulomb repulsive (and the averaged Coulomb repulsive
 potential between electrons), potential between electrons),
 it was rather easy it was rather easy
 to reproduce the atomic to reproduce the atomic
 magic numbers, magic numbers,
 (Z = 2, 10, 18, 36, 54, 86). (Z = 2, 10, 18, 36, 54, 86).
 (See the previous page of the present seminar, (See the previous page of the present seminar,
 "Microscopic World -2-:
3-3: The Periodic Law of Elements"). "Microscopic World -2-:
3-3: The Periodic Law of Elements").

 It was, however, very difficult It was, however, very difficult
 to find out the average potential to find out the average potential
 which can reproduce which can reproduce
 the nuclear magic numbers, the nuclear magic numbers,
 Z Z
 or N or N
 = 2, 8, 20, 28, 50, 82, 126. = 2, 8, 20, 28, 50, 82, 126.

|
 [Mayer-Jensen's shell Model] [Mayer-Jensen's shell Model]

 In 1949, M. G. Mayer In 1949, M. G. Mayer
 (USA, 1906 - 72) (USA, 1906 - 72)
 and H. D. Jensen and H. D. Jensen
 (Germany, 1907 - 73) et al. (Germany, 1907 - 73) et al.
 independently proposed independently proposed
 an average potential an average potential
 which can reproduce the which can reproduce the
 nuclear magic numbers. nuclear magic numbers.
 Thereby the shell structure Thereby the shell structure
 in nuclei in nuclei
 was theoretically was theoretically
 established established
 and it became and it became
 a firm foundation of a firm foundation of
 the Nuclear Structure Theory. the Nuclear Structure Theory.

 The average potential The average potential
 proposed by Mayer, Jensen et al. proposed by Mayer, Jensen et al.
 includes a very special term includes a very special term
 called LS-potential. called LS-potential.
 Because of this, Because of this,
 Mayer-Jensen's shell model Mayer-Jensen's shell model
 is sometimes called is sometimes called
 the jj-coupling shell model. the jj-coupling shell model.
 The details on the LS-potential The details on the LS-potential
 are somewhat expertized discussions, are somewhat expertized discussions,
 so that we omit them here. so that we omit them here.

 In the following figure, In the following figure,
 the single-particle levels the single-particle levels
 in both the models, in both the models,
 the
nuclear shell model the
nuclear shell model
 and the atomic shell model,
and the atomic shell model,
 are compared. are compared.

|
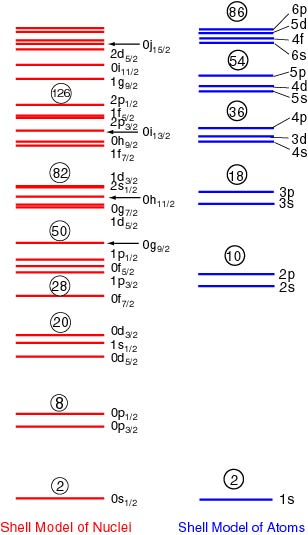
|
 [The Energy Levels [The Energy Levels
 in the Nuclear and Atomic Shell Models] in the Nuclear and Atomic Shell Models]

 The
red levels The
red levels
 show the single-particle levels show the single-particle levels
 in the Mayer-Jensen shell model in the Mayer-Jensen shell model
 for nuclei and for nuclei and
 the
blue levels the
blue levels
 are those in the atomic are those in the atomic
 shell model. shell model.

 In each of the
red levels, In each of the
red levels,
 specified numbers of specified numbers of
 protons and neutrons protons and neutrons
 are filled one by one are filled one by one
 from the bottom, from the bottom,
 and in
blue levels and in
blue levels
 electrons are filled. electrons are filled.
 The circled numbers The circled numbers
 indicate the particle number indicate the particle number
 which can be filled up which can be filled up
 to the levels. to the levels.
 They are just the They are just the
 magic numbers. magic numbers.

|
 Top
Top
|
|

 Go back to
the Top of Part 2. Go back to
the Top of Part 2.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|