
 Top of Part 2
Top of Part 2

 Last page
Last page

 Next page
Next page
  Top of Part 2
Top of Part 2
  Last page
Last page
  Next page
Next page
|
2-2: The Number of Electrons in an Atom |
 As learned in Part 1, As learned in Part 1,
 it turned out it turned out
 that an electron that an electron
 exists in an atom. exists in an atom.

 Are there a fixed number Are there a fixed number
 of electrons of electrons
 in each atom? in each atom?
 The answer to this question The answer to this question
 was given was given
 by the experiment by the experiment
 of the scattering of the scattering
 of X rays from an atom. of X rays from an atom.
 Let's explain Let's explain
 briefly below. briefly below.

|
 [The Property of X Rays] [The Property of X Rays]


 In 1895, In 1895,
 X rays were discovered X rays were discovered
 by W. C. Roentgen by W. C. Roentgen
 (Germany, 1845 - 1923), (Germany, 1845 - 1923),
 while he was doing while he was doing
 experiment experiment
 on vacuum discharge. on vacuum discharge.
 He found He found
 that a kind of "rays" that a kind of "rays"
 which had extremely which had extremely
 high penetrability high penetrability
 were emitted were emitted
 from a Crookes tube from a Crookes tube
 covered by a sheet covered by a sheet
 of thick black paper, of thick black paper,
 and it made some and it made some
 of fluorescent materials of fluorescent materials
 at a distant place at a distant place
 in the room fluoresce in the room fluoresce
 (emit light), (emit light),
 and it exposed and it exposed
 photographic plates photographic plates
 in a drawer of a desk. in a drawer of a desk.
 It was clear It was clear
 that this phenomena that this phenomena
 is not due to is not due to
 the cathode rays, the cathode rays,
 because the cathode rays because the cathode rays
 could not could not
 penetrate a thick penetrate a thick
 paper or a glass. paper or a glass.
 This "rays" were named This "rays" were named
 X rays for X rays for
 the reason that the reason that
 they were unknown. they were unknown.

 The reason The reason
 why X rays come why X rays come
 out of a Crookes tube out of a Crookes tube
 is because the cathode is because the cathode
 rays (electrons) rays (electrons)
 with a very high speed with a very high speed
 collide with glass tube collide with glass tube
 or nearby metal. or nearby metal.
 According to the theory According to the theory
 of electromagnetism, of electromagnetism,
 when a charged when a charged
 particle suddenly accelerate particle suddenly accelerate
 or decelerate, or decelerate,
 it radiates it radiates
 an electromagnetic an electromagnetic
 wave (or light). wave (or light).
 In the vacuum discharge In the vacuum discharge
 tube, a large number of tube, a large number of
 high speed electrons high speed electrons
 collide against collide against
 the glass tube or metal the glass tube or metal
 and are enforced to stop. and are enforced to stop.
 This is sudden This is sudden
 deceleration. deceleration.
 Then an intensive Then an intensive
 electromagnetic wave electromagnetic wave
 is emitted. is emitted.
 This is the reason This is the reason
 of the radiation of the radiation
 of X rays. of X rays.

 The evidence that The evidence that
 X rays are X rays are
 electromagnetic waves electromagnetic waves
 were shown by a were shown by a
 diffraction diffraction
 phenomenon that occurred phenomenon that occurred
 when it passed when it passed
 through a very thin slit. through a very thin slit.
 This phenomenon This phenomenon
 was quite similar was quite similar
 to the case of to the case of
 an ordinary light. an ordinary light.
 A polarization phenomenon A polarization phenomenon
 was confirmed as well. was confirmed as well.
 These phenomena showed These phenomena showed
 that X rays are that X rays are
 electromagnetic waves electromagnetic waves
 like an ordinary light. like an ordinary light.

|
 [Scattering of X Rays [Scattering of X Rays
 by an Atom, by an Atom,
 the Number of Electrons] the Number of Electrons]

 An atom is electrically An atom is electrically
 neutral. neutral.
 We know on the other hand We know on the other hand
 that an atom contains that an atom contains
 electrons electrons
 which are negatively charged. which are negatively charged.
 Accordingly there must Accordingly there must
 exist "something" exist "something"
 with positive charge with positive charge
 which cancels the negative which cancels the negative
 charge of the electrons. charge of the electrons.
 The mass of electron The mass of electron
 is quite light, is quite light,
 i.e. i.e.
 about 1/1800 about 1/1800
 of that of the lightest atom, of that of the lightest atom,
 the hydrogen atom. the hydrogen atom.
 This means that This means that
 the above "something" the above "something"
 with positive charge with positive charge
 must carry almost all must carry almost all
 of the mass of an atom. of the mass of an atom.
 You should keep You should keep
 this in mind. this in mind.

 As mentioned above, As mentioned above,
 it was clarified it was clarified
 that X rays that X rays
 are electromagnetic waves. are electromagnetic waves.
 If X rays are If X rays are
 irradiated to an atom, irradiated to an atom,
 their electric fields their electric fields
 exert forces exert forces
 on the charged objects on the charged objects
 in it (electrons in it (electrons
 or "something" or "something"
 in the atom) in the atom)
 and oscillate them. and oscillate them.
 An oscillating charged An oscillating charged
 object object
 which repeats acceleration which repeats acceleration
 and/or deceleration and/or deceleration
 rapidly emits electromagnetic rapidly emits electromagnetic
 waves around. waves around.
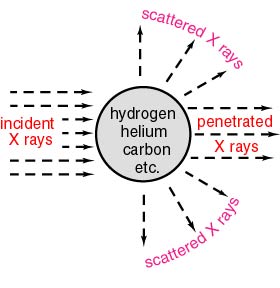
 Thus the incident Thus the incident
 X rays are scattered X rays are scattered
 in various directions in various directions
 as schematically shown as schematically shown
 in the following figure. in the following figure.

|
|
 According to the According to the
 theory of electromagnetism, theory of electromagnetism,
 a charged particle a charged particle
 moving with an acceleration moving with an acceleration
 emits an electromagnetic emits an electromagnetic
 wave whose strength wave whose strength
 is proportional is proportional
 to the square to the square
 of the acceleration. of the acceleration.
 The acceleration The acceleration
 of a particle of a particle
 is obtained by is obtained by
 dividing the force acting dividing the force acting
 on the particle by the mass. on the particle by the mass.
 Therefore the strength Therefore the strength
 of the emitted of the emitted
 electromagnetic wave electromagnetic wave
 is inversely proportional is inversely proportional
 to the square of the mass. to the square of the mass.
 Hence the strength Hence the strength
 of the electromagnetic wave of the electromagnetic wave
 emitted from the "something" emitted from the "something"
 is 1/1000000 less than is 1/1000000 less than
 what is emitted what is emitted
 from an electron, from an electron,
 so that it so that it
 is negligible. is negligible.
 Namely, we can consider that Namely, we can consider that
 X rays are scattered X rays are scattered
 only by the electrons only by the electrons
 in the atom. in the atom.

|
 [The Number of [The Number of
 Electrons in an Atom] Electrons in an Atom]

 Experiments to illuminate X rays Experiments to illuminate X rays
 on various atoms on various atoms
 were carried out were carried out
 as schematically drawn as schematically drawn
 in the above figure. in the above figure.
 In these experiments, In these experiments,
 a part of the incident a part of the incident
 X rays was scattered. X rays was scattered.
 The remaining X rays The remaining X rays
 would penetrate the atoms, would penetrate the atoms,
 and the strength and the strength
 of the penetrated X of the penetrated X
 rays would rays would
 be weakened be weakened
 (or decreased) (or decreased)
 by the scattered X rays. by the scattered X rays.
 This decreasing rate This decreasing rate
 depends on the number depends on the number
 of electrons of electrons
 in the atom. in the atom.
 By investigating this By investigating this
 in detail, in detail,
 the number of electrons the number of electrons
 could roughly be estimated. could roughly be estimated.
 The results told The results told
 that the number that the number
 of electrons of electrons
 in hydrogen atom in hydrogen atom
 is 1 and in helium 2, is 1 and in helium 2,
 and so on. and so on.
 People have known People have known
 that the number that the number
 of electrons in an atom of electrons in an atom
 is about a half is about a half
 of the atomic weight of the atomic weight
 in general. in general.

|
 Top
Top
|
|

 Go back to
the top page of Part 2. Go back to
the top page of Part 2.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|