
 Top of Part 2
Top of Part 2

 Last page
Last page

 Next page
Next page
  Top of Part 2
Top of Part 2
  Last page
Last page
  Next page
Next page
|
2-3: Scattering of Alpha Particle by an Atom |
 Let us summarize Let us summarize
 the properties of atom the properties of atom
 that were made clear that were made clear
 up to the preceding section. up to the preceding section.

|
 (1) (1) |
 The radius of atom The radius of atom
 is approximately is approximately
 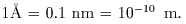

|
|
 (2) (2) |
 An atom includes An atom includes
 electrons, electrons,
 each of which has each of which has
 a negative a negative
 charge -e, charge -e,
 where e where e
 is the elementary charge. is the elementary charge.
 The total mass The total mass
 of the electrons of the electrons
 in an atom is much smaller in an atom is much smaller
 than that of the whole atom. than that of the whole atom.
 The number of electrons The number of electrons
 is about a half of is about a half of
 the atomic weight. the atomic weight.

|
|
 (3) (3) |
 An atom is An atom is
 electrically neutral. electrically neutral.
 So that it includes So that it includes
 "something" whose "something" whose
 charge may cancel charge may cancel
 the charge of all the charge of all
 the electrons. the electrons.
 This "something" must This "something" must
 carry the whole carry the whole
 atomic mass. atomic mass.

|
 What is the internal What is the internal
 structure of such an atom? structure of such an atom?
 In order to study In order to study
 the structure of the structure of
 an invisible atom, an invisible atom,
 it is better to collide it is better to collide
 an appropriate projectile an appropriate projectile
 against the atom against the atom
 and to look and to look
 at the reaction. at the reaction.
 The
alpha rays
The
alpha rays
 were considered were considered
 to be suitable to be suitable
 for this purpose. for this purpose.

|
 [The True Nature of the
Alpha Rays] [The True Nature of the
Alpha Rays]

 Before colliding Before colliding
 the alpha rays the alpha rays
 against an atom, against an atom,
 we have to know we have to know
 what the true nature what the true nature
 of the alpha rays. of the alpha rays.

 The alpha rays The alpha rays
 are beams emitted are beams emitted
 at very high speeds at very high speeds
 from uranium and radium. from uranium and radium.
 They were considered They were considered
 to be collections to be collections
 of the alpha particles of the alpha particles
 charged positively. charged positively.

 First, First,
 the charge-to-mass ratio the charge-to-mass ratio
 of the alpha particle, of the alpha particle,
 Q/M, Q/M,
 was measured was measured
 by curving its orbit by curving its orbit
 in electric and in electric and
 magnetic fields, magnetic fields,
 where Q is the charge where Q is the charge
 of the alpha particle of the alpha particle
 and M the mass. and M the mass.
 Thereby it was seen Thereby it was seen
 that the charge-to-mass that the charge-to-mass
 ratio of the alpha particle ratio of the alpha particle
 was 1/2 of that was 1/2 of that
 of the hydrogen ion. of the hydrogen ion.
 The measurement method The measurement method
 is almost the same as is almost the same as
 the case of electron shown the case of electron shown
 on the page on the page
 1-7-A:
Measurement of the Charge-to-Mass Ratio of Cathode Rays
.
1-7-A:
Measurement of the Charge-to-Mass Ratio of Cathode Rays
.
 But you should note But you should note
 that, that,
 while the charge of electron while the charge of electron
 is negative, is negative,
 that of the that of the
 alpha particle alpha particle
 is positive. is positive.

 Next, the amount Next, the amount
 of the charge of of the charge of
 the alpha particle was the alpha particle was
 measured. measured.
 On the first step On the first step
 for this purpose, for this purpose,
 the number of the number of
 the alpha particles the alpha particles
 emitted in a unit solid-angle emitted in a unit solid-angle
 within a unit time within a unit time
 was measured was measured
 by using the system by using the system
 shown in shown in
 Fig. (A) Fig. (A)
 The
scintillation The
scintillation

 was used for the detector: was used for the detector:
 Namely, Namely,
 the number of the the number of the
 emission of light, emission of light,
 i.e. luminescence, i.e. luminescence,
 which was emitted which was emitted
 from the fluorescent material put from the fluorescent material put
 on the window on the window
 of the detector of the detector
 when being hit when being hit
 by a charged particle, by a charged particle,
 was counted. was counted.

|
|
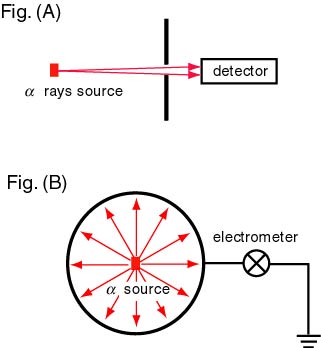
 On the second step, On the second step,
 the total charge the total charge
 of the alpha rays of the alpha rays
 radiated in a unit time radiated in a unit time
 from the same source from the same source
 is measured is measured
 by using an apparatus by using an apparatus
 shown in the above figure, shown in the above figure,
 Fig. (B). Fig. (B).
 Dividing the resultant Dividing the resultant
 total charge by total charge by
 the number of the number of
 the alpha particles, the alpha particles,
 they obtained they obtained
 the value of electric charge the value of electric charge
 per particle. per particle.
 Thereby Thereby

 Charge of an alpha particle = Charge of an alpha particle =
 +2e +2e
 (e = the elementary charge) (e = the elementary charge)

 was clarified. was clarified.
 An alpha particle An alpha particle
 is therefore is therefore
 speculated to be speculated to be
 a helium ion, a helium ion,
 i.e. the helium atom i.e. the helium atom
 having lost having lost
 two electrons. two electrons.

 It was later confirmed It was later confirmed
 by E. Rutherford by E. Rutherford
 (UK, 1871 - 1937) (UK, 1871 - 1937)
 and and
 T. Royds T. Royds
 that this speculation that this speculation
 was correct (1908). was correct (1908).
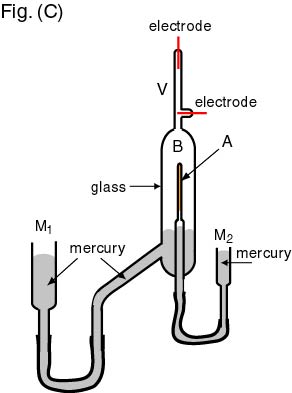
 The schematic drawing The schematic drawing
 of the apparatus of the apparatus
 used for this purpose used for this purpose
 is shown in the is shown in the
 following figure, following figure,
 Fig. (C). Fig. (C).

|
|
 In the above apparatus, In the above apparatus,
 a radiative material a radiative material
 (gas) is enclosed (gas) is enclosed
 in a very thin in a very thin
 glass tube A glass tube A
 under a pressure under a pressure
 applied by the mercury applied by the mercury
 M2 M2
 at the center at the center
 of a vacuum glass of a vacuum glass
 container B. container B.
 The alpha particles The alpha particles
 emitted from the radiative gas emitted from the radiative gas
 penetrate the thin glass penetrate the thin glass
 to be accumulated to be accumulated
 in the outside glass in the outside glass
 container B. container B.

 After leaving After leaving
 this apparatus this apparatus
 for several days, for several days,
 the mercury M1 the mercury M1
 was lifted up was lifted up
 so that the gas accumulated so that the gas accumulated
 in the container in the container
 B was led up B was led up
 into an upper narrow tube into an upper narrow tube
 V. V.
 They made a discharge They made a discharge
 by applying by applying
 a high voltage a high voltage
 between the electrodes, between the electrodes,
 and observed the spectrum and observed the spectrum
 which was exactly which was exactly
 the same as the same as
 that of the helium atom. that of the helium atom.
 Thus they confirmed Thus they confirmed
 that the gas accumulated that the gas accumulated
 in the container in the container
 B was helium gas. B was helium gas.

 It was confirmed It was confirmed
 that an alpha particle that an alpha particle
 came from A to B came from A to B
 captured two units captured two units
 of elementary charge of elementary charge
 from environment from environment
 to become a helium atom. to become a helium atom.
 This means This means
 that the alpha particle that the alpha particle
 is a doubly ionized is a doubly ionized
 helium atom. helium atom.

 For reference, For reference,
 they put helium gas they put helium gas
 in the thin tube A in the thin tube A
 and leave it and leave it
 for several days, for several days,
 but they could find but they could find
 no helium gas no helium gas
 in the outer container in the outer container
 B B
 at all; at all;
 namely, namely,
 an alpha particle an alpha particle
 can penetrate can penetrate
 the thin glass wall the thin glass wall
 of the tube A, of the tube A,
 but a helium atom cannot. but a helium atom cannot.
 Let's think why it is so. Let's think why it is so.
|

 [Scattering of the Alpha [Scattering of the Alpha
 Rays by an Atom] Rays by an Atom]

 The experiment of The experiment of
 the scattering of alpha rays the scattering of alpha rays
 by an atom were carried out by an atom were carried out
 by by
 H. W. Geiger H. W. Geiger
 (Germany, 1882 -1945) and (Germany, 1882 -1945) and
 E. Marsden E. Marsden
 (UK, 1889 - 1970) (UK, 1889 - 1970)
 under Rutherford's under Rutherford's
 leadership (1909). leadership (1909).
 They made alpha rays They made alpha rays
 emitted from radium collide emitted from radium collide
 against a thin metal against a thin metal
 (gold or silver) foil. (gold or silver) foil.
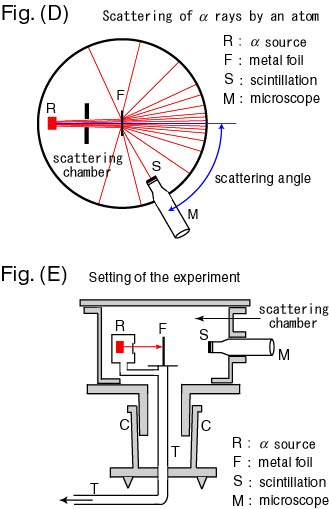
 A schematic graph A schematic graph
 of the experiment of the experiment
 is shown in is shown in
 Fig. (D) Fig. (D)
 and the details and the details
 of the apparatus of the apparatus
 are in are in
 Fig. (E). Fig. (E).

 In the vacuum scattering In the vacuum scattering
 chamber in chamber in
 Fig. (D), Fig. (D),
 the alpha particles the alpha particles
 coming out of the source R coming out of the source R
 hit the metal foil F hit the metal foil F
 at the center and at the center and
 they are scattered they are scattered
 in various directions. in various directions.
 These scattered particles These scattered particles
 are observed through are observed through
 their scintillations their scintillations
 with the microscope M. with the microscope M.
 Thereby Thereby
 it is measured it is measured
 what rate of particle number is what rate of particle number is
 scattered in a given scattered in a given
 solid angle. solid angle.

 The results obtained The results obtained
 by Geiger and Marsden by Geiger and Marsden
 showed that showed that
 the almost all alpha particles the almost all alpha particles
 go straight go straight
 in the forward angle, in the forward angle,
 but but
 a few number of particles a few number of particles
 are occasionally are occasionally
 scattered in a scattered in a
 very large angle very large angle
 like 90 degrees like 90 degrees
 or larger. or larger.

|
|
 Top
Top
|
|

 Go back to
the top page of Part 2. Go back to
the top page of Part 2.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|