
 Top of Part 2
Top of Part 2

 Last page
Last page

 Next page
Next page
  Top of Part 2
Top of Part 2
  Last page
Last page
  Next page
Next page
|
2-5: Rutherford's Atomic Model |
 As discussed As discussed
 in the preceding page, in the preceding page,
 we should consider that we should consider that
 Thomson's raisin bread model Thomson's raisin bread model
 (plum pudding model) (plum pudding model)
 was unsuccessful, was unsuccessful,
 because it could because it could
 not explain the results not explain the results
 of the alpha particle of the alpha particle
 scattering experiment scattering experiment
 of of
 Geiger and Marsden. Geiger and Marsden.

 Let us look back Let us look back
 upon the experimental results; upon the experimental results;
 we can summarize them we can summarize them
 as follows: as follows:

 [The Results of [The Results of
 Geiger and Marsden's Geiger and Marsden's
 experiment] experiment]

|
 (1) (1) |
 Almost all the incident Almost all the incident
 alpha particles alpha particles
 go straight and go straight and
 are scarcely scattered. are scarcely scattered.

| |
 (2) (2) |
 Only occasionally Only occasionally
 such a large-angle such a large-angle
 scattering through scattering through
 an angle greater an angle greater
 than 90 degrees than 90 degrees
 or near 180 degrees or near 180 degrees
 occurs. occurs.

| |
 (3) (3) |
 The scattering rate The scattering rate
 (or probability) (or probability)
 depends on the atomic depends on the atomic
 weight of the target; weight of the target;
 the more the atomic weight, the more the atomic weight,
 the larger the probability. the larger the probability.

|


|
 [The Rutherford Model of the Nuclear Atom] [The Rutherford Model of the Nuclear Atom]

 Considering the results Considering the results
 of Geiger and Marsden's of Geiger and Marsden's
 experiment experiment
 and the failure and the failure
 of Thomson's atomic model, of Thomson's atomic model,
 E. Rutherford E. Rutherford
 (UK, 1871 - 1937) (UK, 1871 - 1937)
 proposed a model proposed a model
 in which in which
 the electric charge +Ze the electric charge +Ze
 in an atom in an atom
 is not distributed is not distributed
 over the whole area over the whole area
 of the atom of the atom
 but concentrates but concentrates
 in a small area in a small area
 (1911). (1911).

 He thought as follows: He thought as follows:
 The charge +Ze The charge +Ze
 is localized to be is localized to be
 a cluster or a group a cluster or a group
 and the alpha particle and the alpha particle
 is scattered by is scattered by
 Coulomb's repulsive force, Coulomb's repulsive force,
 (C. A. de Coulomb: (C. A. de Coulomb:
 France, 1736 - 1806). France, 1736 - 1806).
 Namely, his idea is that Namely, his idea is that
 the large-angle scattering the large-angle scattering
 of the alpha particle of the alpha particle
 is brought about is brought about
 by a single scattering by a single scattering
 due to Coulomb's repulsive force due to Coulomb's repulsive force
 between the charge between the charge
 of the alpha particle, of the alpha particle,
 +2e, and +2e, and
 that of the cluster, that of the cluster,
 +Ze. +Ze.
 The cluster is called The cluster is called
 atomic nucleus atomic nucleus
 or simply nucleus. or simply nucleus.
 Rutherford's atomic model Rutherford's atomic model
 is often called is often called
 Rutherford model Rutherford model
 or sometimes or sometimes
 Rutherford model Rutherford model
 of the nuclear atom. of the nuclear atom.

 A schematic image A schematic image
 of the Rutherford model of the Rutherford model
 is shown in the following is shown in the following
 figure, figure,
 where a black big ball where a black big ball
 at the center at the center
 is the nucleus and is the nucleus and
 small red points moving small red points moving
 around the nucleus around the nucleus
 are electrons. are electrons.

|

|
 The Rutherford Model of the Nuclear Atom
The Rutherford Model of the Nuclear Atom

 A black big ball A black big ball
 at the center at the center
 is the nucleus and is the nucleus and
 small red points moving small red points moving
 around the nucleus around the nucleus
 are electrons. are electrons.

|
 In 1903, In 1903,
 H. Nagaoka H. Nagaoka
 (Japan, 1865 - 1950) (Japan, 1865 - 1950)
 proposed a ``saturnian model'', proposed a ``saturnian model'',
 which was referred to which was referred to
 in Rutherford's paper. in Rutherford's paper.
 You should note You should note
 that the Nagaoka's model that the Nagaoka's model
 had been proposed had been proposed
 several years several years
 before Rutherford's model. before Rutherford's model.

|
 [Rutherford Scattering] [Rutherford Scattering]

 Rutherford investigated Rutherford investigated
 whether the results whether the results
 of the experiment of the experiment
 of the alpha particle scattering of the alpha particle scattering
 could be explained well could be explained well
 by the Rutherford model by the Rutherford model
 of the nuclear atom. of the nuclear atom.
 He derived the famous He derived the famous
 Rutherford scattering formula Rutherford scattering formula
 standing on the viewpoint standing on the viewpoint
 of the Rutherford model of the Rutherford model
 and he found and he found
 that the results that the results
 of this formula of this formula
 fit well to fit well to
 the experimental data the experimental data
 (1911). (1911).
 Thus the firm position Thus the firm position
 of the Rutherford model of the Rutherford model
 was established. was established.

 Rutherford assumed Rutherford assumed
 that the total positive that the total positive
 charge in an atom, +Ze, charge in an atom, +Ze,
 concentrates on the central concentrates on the central
 point of the atom, point of the atom,
 i.e., the nucleus, i.e., the nucleus,
 and the incident alpha particle and the incident alpha particle
 is scattered is scattered
 with a repulsive Coulomb with a repulsive Coulomb
 force exerted force exerted
 by this nuclear point charge. by this nuclear point charge.
 (See the following (See the following
 figure.) figure.)

|

|
 This type of scattering This type of scattering
 by the Coulomb force by the Coulomb force
 is usually called is usually called
 Coulomb scattering or Coulomb scattering or
 Rutherford scattering. Rutherford scattering.
 (See the above (See the above
 figure.) figure.)

 The detailed formulation The detailed formulation
 on Rutherford scattering on Rutherford scattering
 will be given will be given
 in the following other page. in the following other page.
 If you feel it difficult If you feel it difficult
 to understand, to understand,
 skip it, skip it,
 and accept the final and accept the final
 Rutherford scattering formula. Rutherford scattering formula.

 2-5-A:
Rutherford Scattering
2-5-A:
Rutherford Scattering

|
 As shown in As shown in
 the above figure, the above figure,
 the distance between the distance between
 the incident line the incident line
 and the nucleus and the nucleus
 is the impact parameter is the impact parameter
 b. b.

 If b = 0, If b = 0,
 it is head-on collision, it is head-on collision,
 where the incident alpha particle where the incident alpha particle
 would recoil would recoil
 in the direction in the direction
 of 180 degrees. of 180 degrees.
 If b If b
 is small, is small,
 the trajectory the trajectory
 would be curved largely. would be curved largely.
 If b is large If b is large
 and the particle and the particle
 goes along a path goes along a path
 far from the nucleus, far from the nucleus,
 then the repulsive force then the repulsive force
 exerted by the nucleus exerted by the nucleus
 is weak and is weak and
 the trajectory would not the trajectory would not
 curve so much. curve so much.

 Namely, Namely,
 the trajectory is determined the trajectory is determined
 by the impact parameter only by the impact parameter only
 for a constant incident speed for a constant incident speed
 v0, v0,
 This feature are shown This feature are shown
 in the following graph in the following graph
 which is drawn which is drawn
 by using the numerical data by using the numerical data
 obtained from the solution obtained from the solution
 discussed on the page discussed on the page
 (2-5-A).
(2-5-A).


|
 [Angular Distribution [Angular Distribution
 of Rutherford Scattering] of Rutherford Scattering]

 As seen in the above figure, As seen in the above figure,
 if the incident line if the incident line
 is close to the nucleus is close to the nucleus
 of the target atom of the target atom
 or if the impact parameter or if the impact parameter
 is small, is small,
 the trajectory the trajectory
 of the alpha particle of the alpha particle
 would be largely would be largely
 curved and curved and
 it would be scattered it would be scattered
 backward. backward.
 In contrast, In contrast,
 if the impact parameter if the impact parameter
 is large, is large,
 then the alpha particle then the alpha particle
 goes through far goes through far
 from the nucleus and from the nucleus and
 the trajectory the trajectory
 does not curve so much. does not curve so much.

 In the realistic experiment, In the realistic experiment,
 the number of the incident the number of the incident
 alpha particles alpha particles
 in a unit area in a unit area
 is almost constant is almost constant
 independently of the independently of the
 impact parameter. impact parameter.
 The number of the The number of the
 incident particles incident particles
 in a unit area in a unit area
 and a unit time, and a unit time,
 N, N,
 is called the is called the
 intensity intensity
 of the incident alpha rays. of the incident alpha rays.
 We may consider that We may consider that
 the intensity N the intensity N
 of the incident alpha rays of the incident alpha rays
 is constant. is constant.

 It is called It is called
 the angular distribution the angular distribution
 what rate among what rate among
 the many incident particles the many incident particles
 is scattered in the direction is scattered in the direction
 of a certain angle. of a certain angle.
 Strictly speaking, Strictly speaking,
 the angular distribution the angular distribution
 or or
 differential scattering differential scattering
 cross section cross section
 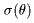
 is defined by dividing is defined by dividing
 the number of the number of
 the particles scattered the particles scattered
 in a unit solid angle in a unit solid angle
 of a certain scattering angle of a certain scattering angle
 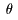
 in a unit time in a unit time
 by the intensity N. by the intensity N.

 Rutherford obtained Rutherford obtained
 the differential scattering the differential scattering
 cross section cross section
 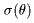
 on the basis of on the basis of
 Newtonian mechanics Newtonian mechanics
 (1911). (1911).
 The details of deriving The details of deriving
 the cross section the cross section
 are explained on the page are explained on the page
 2-5-A
2-5-A

 The result is shown The result is shown
 in the following formula, in the following formula,


 This Rutherford's formula This Rutherford's formula
 was well fit to experiment. was well fit to experiment.
 Thus the validity Thus the validity
 of Rutherford's model of Rutherford's model
 of the nuclear atom of the nuclear atom
 has been established. has been established.

|
 As stated above, As stated above,
 Rutherford derived Rutherford derived
 the angular distribution the angular distribution
 of the alpha particle scattering of the alpha particle scattering
 on the basis on the basis
 of Newtonian mechanics. of Newtonian mechanics.
 Newtonian mechanics Newtonian mechanics
 is sometimes called is sometimes called
 classical mechanics, classical mechanics,
 and it was later and it was later
 clarified that clarified that
 Newtonian mechanics Newtonian mechanics
 is not always correct is not always correct
 in the microscopic world, in the microscopic world,
 i.e. i.e.
 the world of the world of
 atoms and atomic nuclei. atoms and atomic nuclei.
 It has been made clear It has been made clear
 some years later that some years later that
 the correct theory the correct theory
 to describe such to describe such
 a microscopic world a microscopic world
 is quantum mechanics. is quantum mechanics.

 It has however been It has however been
 clarified that, clarified that,
 as long as Rutherford scattering as long as Rutherford scattering
 is concerned, is concerned,
 both classical mechanics both classical mechanics
 and quantum mechanics and quantum mechanics
 give the same angular distribution. give the same angular distribution.

|
 [The Size of the Charge [The Size of the Charge
 of Atomic Nucleus] of Atomic Nucleus]

 The angular distribution The angular distribution
 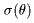
 derived by derived by
 Rutherford was Rutherford was
 very successful not only very successful not only
 in the angular dependence in the angular dependence
 but in a by-product. but in a by-product.
 As seen in the above As seen in the above
 Rutherford formula, Rutherford formula,
 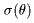
 contains a factor Z. contains a factor Z.
 By measuring By measuring
 this scattering this scattering
 probability precisely, probability precisely,
 we can obtain we can obtain
 the value of Z. the value of Z.

 The quantity Z The quantity Z
 is the number of the electrons is the number of the electrons
 surrounding the nucleus in an atom. surrounding the nucleus in an atom.
 Before Rutherford's formula, Before Rutherford's formula,
 the value of Z the value of Z
 had only been known had only been known
 to be about a half to be about a half
 of the atomic weight. of the atomic weight.
 However, precise measurements However, precise measurements
 of the angular distribution of the angular distribution
 of Rutherford scattering of Rutherford scattering
 have shown the precise value have shown the precise value
 of the charge of the charge
 of the atomic nucleus of the atomic nucleus
 and also the number and also the number
 of electrons in an atom. of electrons in an atom.
 Consequently, Consequently,
 it was made clear it was made clear
 that Z that Z
 is exactly equal is exactly equal
 to the atomic number. to the atomic number.

|
 Top
Top
|
|

 Go back to
the top page of Part 2. Go back to
the top page of Part 2.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|