
 Top of Part 1
Top of Part 1

 Last page
Last page

 Top of Part 2
Top of Part 2
  Top of Part 1
Top of Part 1
  Last page
Last page
  Top of Part 2
Top of Part 2
|
1-4: The Constituents of Nuclei, Summary |
 [The Constituents of Nuclei] [The Constituents of Nuclei]

 Up to the 1930s, Up to the 1930s,
 people had thought that people had thought that
 the atomic nucleus consists the atomic nucleus consists
 of protons and of protons and
 electrons. electrons.
 For example, they considered For example, they considered
 that the nitrogen 14 of that the nitrogen 14 of
 the atomic number Z = 7 the atomic number Z = 7
 consists of 14 protons and consists of 14 protons and
 7 electrons. 7 electrons.
 This idea appeared quite This idea appeared quite
 plausible, but it was made plausible, but it was made
 clear that it contained clear that it contained
 serious difficulties serious difficulties
 as explained on the as explained on the
 last page. last page.

 Just after Just after
 the discovery of the neutron the discovery of the neutron
 by Chadwick, by Chadwick,
 D. Ivanenko (Soviet Union) D. Ivanenko (Soviet Union)
 and and
 W. K. Heisenberg W. K. Heisenberg
 (Germany, 1901-76) (Germany, 1901-76)
 independently proposed independently proposed
 an idea that an idea that
 "the atomic nucleus consists "the atomic nucleus consists
 of protons and neutrons" (1932). of protons and neutrons" (1932).
 This was nothing else than This was nothing else than
 the first step of the first step of
 Nuclear Physics. Nuclear Physics.
 Hereafter this idea has been Hereafter this idea has been
 established; established;
 namely, a nucleus of an atomic number namely, a nucleus of an atomic number
 Z Z
 is a composite many-particle system is a composite many-particle system
 consisting of consisting of
 Z protons and Z protons and
 N neutrons. N neutrons.

 Accordingly, for example, Accordingly, for example,
 the nitrogen 14 nucleus of the nitrogen 14 nucleus of
 the atomic number 7 the atomic number 7
 consists of 7 protons and consists of 7 protons and
 7 neutrons. 7 neutrons.
 And we can easily understand that And we can easily understand that
 there exist some nuclei (isotopes) there exist some nuclei (isotopes)
 of the same atomic number Z of the same atomic number Z
 but different neutron numbers but different neutron numbers
 N. N.

|
 [The Nucleon] [The Nucleon]

 The proton and the neutron The proton and the neutron
 have almost the same mass. have almost the same mass.
 Different is their electric charges. Different is their electric charges.
 Their currently accepted properties Their currently accepted properties
 are listed in the following Table. are listed in the following Table.


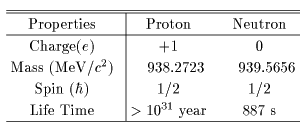
 From these properties, From these properties,
 Heisenberg and Heisenberg and
 E. P. Wigner (US, 1902-95) E. P. Wigner (US, 1902-95)
 thought that these two kinds thought that these two kinds
 of particles are of two of particles are of two
 different states different states
 of the same particle, of the same particle,
 called "nucleon" today. called "nucleon" today.

 We can therefore say, We can therefore say,
 in a word, that in a word, that
 the constituent of the atomic nucleus the constituent of the atomic nucleus
 is nucleon. is nucleon.

|
 [Summary of Part 1] [Summary of Part 1]

 Let us summarize Let us summarize
 what we have learned what we have learned
 in Part 1. in Part 1.

|
 (1) (1) |
 After the success of After the success of
 the Rutherford Model of the Rutherford Model of
 the Nuclear Atom, the Nuclear Atom,
 the existence of the atomic nucleus the existence of the atomic nucleus
 was recognized for the first time. was recognized for the first time.

|
 (2) (2) |
 The mass of the nucleus The mass of the nucleus
 is almost equal to that of is almost equal to that of
 the whole atom. the whole atom.
 Its diameter is about Its diameter is about

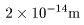 , ,
 which is extremely smaller which is extremely smaller
 than the size of the whole atom, about than the size of the whole atom, about
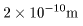 . .

|
 (3) (3) |
 Due to the discovery of Due to the discovery of
 the neutron by Chadwick, the neutron by Chadwick,
 it was clarified that it was clarified that
 the constituents of nuclei the constituents of nuclei
 are protons are protons
 and neutrons and neutrons
 (generically nucleons). (generically nucleons).

|
 Top
Top
|
|
 Go back to
the top page of Part 1. Go back to
the top page of Part 1.

 Go back to
the last page. Go back to
the last page.

 Go to
the top page of Part 2. Go to
the top page of Part 2.
|