
 Top of Part 1
Top of Part 1

 Last page
Last page

 Next page
Next page
  Top of Part 1
Top of Part 1
  Last page
Last page
  Next page
Next page
|
1-4: The Motion of Molecules |
 [Doubt about Atomism] [Doubt about Atomism]

 The kinetic theory The kinetic theory
 of gases of gases
 and and
 statistical mechanics statistical mechanics
 were developed by were developed by
 J. C. Maxwell J. C. Maxwell
 (UK, 1831 - 79) (UK, 1831 - 79)
 and and
 L. Boltzmann L. Boltzmann
 (Germany, 1844 - 1906) (Germany, 1844 - 1906)
 in the 19th century. in the 19th century.
 These theories These theories
 are based are based
 on the theory of atoms on the theory of atoms
 (atomism) (atomism)
 and molecules. and molecules.
 Looking at the success Looking at the success
 of these theories, of these theories,
 they thought that the theories of they thought that the theories of
 the atomism and the atomism and
 of molecules of molecules
 were firmly established. were firmly established.

 However, even However, even
 at the beginning of at the beginning of
 the 20th century, the 20th century,
 some leading physicists some leading physicists
 strongly opposed strongly opposed
 to the ideas of to the ideas of
 atomic atomic
 and molecular theory, and molecular theory,
 insisting that insisting that
 the idea is nothing the idea is nothing
 but a product but a product
 of human imagination of human imagination
 and and
 there had been no there had been no
 experimental proof experimental proof
 of the existence of the existence
 of atoms and molecules, of atoms and molecules,
 although the idea although the idea
 was very convenient. was very convenient.

 What smashed What smashed
 this opposition this opposition
 was the observation was the observation
 of Brownian motion. of Brownian motion.

|
 [Speed of the Molecular Motion] [Speed of the Molecular Motion]

 Molecules in a gas Molecules in a gas
 are always moving are always moving
 rapidly and busily. rapidly and busily.

 J. P. Joule J. P. Joule
 (UK, 1818 - 89) (UK, 1818 - 89)
 estimated the speed estimated the speed
 of a molecule in a gas of a molecule in a gas
 for the first time for the first time
 (1851). (1851).

 He thought that He thought that
 a gas molecule a gas molecule
 in a container in a container
 moves around moves around
 at a high speed at a high speed
 and sometimes collides and sometimes collides
 against the wall against the wall
 of the container. of the container.
 When colliding, When colliding,
 the molecule exerts the molecule exerts
 forces on the wall. forces on the wall.
 The forces are summed up The forces are summed up
 as the pressure as the pressure
 of the gas. of the gas.
 This is Joule's This is Joule's
 basic idea basic idea
 on gaseous pressure on gaseous pressure
 from the viewpoint from the viewpoint
 of the molecular theory. of the molecular theory.

 Standing on this viewpoint, Standing on this viewpoint,
 let us estimate let us estimate
 the molecular speed the molecular speed
 in a gas below. in a gas below.
 To do this, To do this,
 we need some mathematical we need some mathematical
 expressions. expressions.
 Although they are not Although they are not
 so difficult, so difficult,
 let us explain let us explain
 on the other page: on the other page:

 1-4-A:
Speed of Gaseous Molecules
1-4-A:
Speed of Gaseous Molecules

 As seen on the page As seen on the page
 (1-4-A), (1-4-A),
 a molecule in a gas a molecule in a gas
 is moving at an extremely is moving at an extremely
 high speed, high speed,
 almost equal almost equal
 to the speed of sound. to the speed of sound.
 It is generally It is generally
 thought that thought that
 molecules in a gas molecules in a gas
 and a liquid and a liquid
 are moving randomly are moving randomly
 at a very high speed. at a very high speed.

 We cannot watch We cannot watch
 those motions directly, those motions directly,
 but, if we can observe but, if we can observe
 them in any indirect way, them in any indirect way,
 we could confirm we could confirm
 the existence of molecule. the existence of molecule.
 This has been enabled This has been enabled
 by the observation of by the observation of
 Brownian motion. Brownian motion.

|
 [Brownian Motion] [Brownian Motion]

 A botanist A botanist
 R. Brown R. Brown
 (UK, 1773 - 1858) (UK, 1773 - 1858)
 observed irregular observed irregular
 motions of very fine motions of very fine
 particles particles
 which came out of pollens which came out of pollens
 floated on water. floated on water.
 He put pollens He put pollens
 on water, on water,
 where they absorbed where they absorbed
 sufficient water sufficient water
 to be burst into to be burst into
 very fine particles. very fine particles.
 He observed the irregular He observed the irregular
 zigzag motions zigzag motions
 of these particles of these particles
 using a microscope using a microscope
 (1827). (1827).
 He reported that He reported that
 such fine particles such fine particles
 come not only out of come not only out of
 pollens but also pollens but also
 from other substances from other substances
 and they show similar and they show similar
 irregular motions. irregular motions.

 This irregular motion This irregular motion
 was called was called
 Brownian motion Brownian motion
 that is thought that is thought
 to be a rather to be a rather
 universal motion universal motion
 seen not only in such seen not only in such
 a liquid as water a liquid as water
 but also for a particle but also for a particle
 of soot in air. of soot in air.
 It appears to occur It appears to occur
 because because
 the fine particle the fine particle
 is violently colliding is violently colliding
 with molecules of the gas with molecules of the gas
 or the liquid or the liquid
 moving randomly moving randomly
 at high speed. at high speed.
 A. Einstein A. Einstein
 (Germany, USA, 1879 - 1955) (Germany, USA, 1879 - 1955)
 gave a theoretical gave a theoretical
 interpretation interpretation
 of Brownian motion of Brownian motion
 based on the viewpoint based on the viewpoint
 of the molecular theory of the molecular theory
 (1905). (1905).

|


|
 In 1908 - 13, In 1908 - 13,
 J. B. Perrin J. B. Perrin
 (France, 1870 - 1942) (France, 1870 - 1942)
 carried out carried out
 a lot of difficult a lot of difficult
 experiments repeatedly, experiments repeatedly,
 and he was finally and he was finally
 successful in proving successful in proving
 Einstein's Einstein's
 molecular theory. molecular theory.
 Thereby he succeeded Thereby he succeeded
 in estimating in estimating
 the size of the molecule the size of the molecule
 and obtaining the value and obtaining the value
 of Avogadro's constant. of Avogadro's constant.

 Looking at the results Looking at the results
 of Perrin, of Perrin,
 the actual existence the actual existence
 of atoms of atoms
 and molecules and molecules
 has widely been has widely been
 recognized and accepted. recognized and accepted.

|
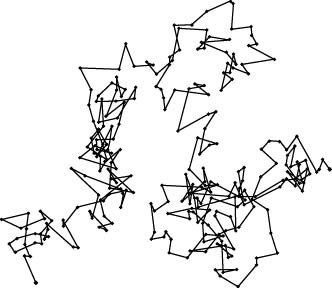
|
An Example of Brownian Motion observed by Perrin.
observed by Perrin.

 The position The position
 of a fine particle of a fine particle
 of olibanum of olibanum
 is recorded is recorded
 for every constant for every constant
 interval. interval.
 The actual motion The actual motion
 is three-dimensional, is three-dimensional,
 but the figure shows but the figure shows
 the position the position
 projected on a plane. projected on a plane.

|
 Top
Top
|
|

 Go back to
the top page of Part 1. Go back to
the top page of Part 1.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|