| |
 People realized People realized
 that atoms that atoms
 exists surely. exists surely.
 Then they questioned Then they questioned
 what the size what the size
 of an atom is. of an atom is.

 According to various According to various
 experiments, experiments,
 Avogadro's constant Avogadro's constant
 has turned out to be has turned out to be




 Since a solid Since a solid
 like a metal block like a metal block
 is tightly filled is tightly filled
 with atoms as shown with atoms as shown
 in the following figure, in the following figure,
 we can easily estimate we can easily estimate
 the size of the atom. the size of the atom.



 Suppose the radius of atom Suppose the radius of atom
 to be r cm. to be r cm.
 Then Then
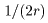
 atoms would line up in 1 cm atoms would line up in 1 cm
 and and

 atoms occupy atoms occupy
 the volume of the volume of

 Suppose the density Suppose the density
 of the metal to be of the metal to be

 Let the molecular Let the molecular
 weight of the metal, weight of the metal,
 which is equal to which is equal to
 the atomic weight the atomic weight
 in the case of metal, in the case of metal,
 be A. be A.
 The volume of The volume of
 1 mol 1 mol
 of the metal of the metal
 is is
 . .
 Therefore the volume Therefore the volume
 occupied by one atom occupied by one atom
 of the metal of the metal
 is obtained by dividing is obtained by dividing

 by Avogadro's constant by Avogadro's constant
 NA. NA.
 Namely, Namely,




 Accordingly, the radius Accordingly, the radius
 of the atom is of the atom is
 considered to be considered to be




 For example, For example,
 the radii of some the radii of some
 solid-state atoms solid-state atoms
 estimated by using estimated by using
 the above formula the above formula
 are listed in the following are listed in the following
 Table. Table.




 From these results, From these results,
 we can presume that we can presume that
 the size of the size of
 an atom is approximately an atom is approximately
 several angstroms several angstroms
 (1 angstrom (1 angstrom
 = 10-8 cm = 10-8 cm
 = 10-10 m). = 10-10 m).

|

 Top of Part 1
Top of Part 1

 Last page
Last page

 Next page
Next page

 Top of Part 1
Top of Part 1

 Last page
Last page

 Next page
Next page
 People realized
People realized
 that atoms
that atoms
 exists surely.
exists surely.
 Then they questioned
Then they questioned
 what the size
what the size
 of an atom is.
of an atom is.

 According to various
According to various
 experiments,
experiments,
 Avogadro's constant
Avogadro's constant
 has turned out to be
has turned out to be




 Since a solid
Since a solid
 like a metal block
like a metal block
 is tightly filled
is tightly filled
 with atoms as shown
with atoms as shown
 in the following figure,
in the following figure,
 we can easily estimate
we can easily estimate
 the size of the atom.
the size of the atom.



 Suppose the radius of atom
Suppose the radius of atom
 to be r cm.
to be r cm.
 Then
Then
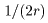
 atoms would line up in 1 cm
atoms would line up in 1 cm
 and
and

 atoms occupy
atoms occupy
 the volume of
the volume of

 Suppose the density
Suppose the density
 of the metal to be
of the metal to be

 Let the molecular
Let the molecular
 weight of the metal,
weight of the metal,
 which is equal to
which is equal to
 the atomic weight
the atomic weight
 in the case of metal,
in the case of metal,
 be A.
be A.
 The volume of
The volume of
 1 mol
1 mol
 of the metal
of the metal
 is
is
 .
.
 Therefore the volume
Therefore the volume
 occupied by one atom
occupied by one atom
 of the metal
of the metal
 is obtained by dividing
is obtained by dividing

 by Avogadro's constant
by Avogadro's constant
 NA.
NA.
 Namely,
Namely,




 Accordingly, the radius
Accordingly, the radius
 of the atom is
of the atom is
 considered to be
considered to be




 For example,
For example,
 the radii of some
the radii of some
 solid-state atoms
solid-state atoms
 estimated by using
estimated by using
 the above formula
the above formula
 are listed in the following
are listed in the following
 Table.
Table.




 From these results,
From these results,
 we can presume that
we can presume that
 the size of
the size of
 an atom is approximately
an atom is approximately
 several angstroms
several angstroms
 (1 angstrom
(1 angstrom
 = 10-8 cm
= 10-8 cm
 = 10-10 m).
= 10-10 m).

 Top
Top

 Go back to
the top page of Part 1.
Go back to
the top page of Part 1.

 Go back to
the last page.
Go back to
the last page.

 Go to
the next page.
Go to
the next page.