
 Top of Part 1
Top of Part 1

 Last page
Last page

 Next page
Next page
  Top of Part 1
Top of Part 1
  Last page
Last page
  Next page
Next page
|
1-6: The Discovery of Elementary Charge |
 At the beginning At the beginning
 of the 19th century, of the 19th century,
 experiments on experiments on
 electrolysis electrolysis
 were frequently were frequently
 carried out. carried out.
 For example, For example,
 it happens it happens
 that, if water is that, if water is
 electrolyzed, electrolyzed,
 oxygen and hydrogen oxygen and hydrogen
 are generated. are generated.

 We put electrolyte solution We put electrolyte solution
 in a container called in a container called
 "electrolytic cell" "electrolytic cell"
 and put and put
 positive and positive and
 negative electrodes in it. negative electrodes in it.
 When we send When we send
 a direct electric current a direct electric current
 between the electrodes, between the electrodes,
 the negative ions the negative ions
 are attracted to the are attracted to the
 positive electrode, positive electrode,
 and the positive ions and the positive ions
 to the negative electrode. to the negative electrode.
 Thus the electolytic solution Thus the electolytic solution
 is decomposed at these is decomposed at these
 electrodes. electrodes.
 This phenomena is This phenomena is
 the electrolysis. the electrolysis.

 In the case of In the case of
 the following figure, the following figure,
 a solution of copper sulfate a solution of copper sulfate
 is to be electrolyzed. is to be electrolyzed.




 [Faraday's Law of Electrolysis]
[Faraday's Law of Electrolysis]

|


|
 M. Faraday M. Faraday
 (UK, 1791 - 1867) (UK, 1791 - 1867)
 repeatedly carried out repeatedly carried out
 experiments concerning experiments concerning
 electrolysis electrolysis
 and discovered and discovered
 the following law the following law
 called Faraday's law called Faraday's law
 of electrolysis of electrolysis
 (1833).: (1833).:

 Here the chemical Here the chemical
 equivalent means equivalent means
 the value obtained the value obtained
 by dividing by dividing
 the atomic weight the atomic weight
 by the valence by the valence
 (or atomic value) (or atomic value)
 of the element. of the element.

|
 [The Valence] [The Valence]

 The valence The valence
 of an atom is of an atom is
 its combining capacity. its combining capacity.
 Namely, it is the number Namely, it is the number
 of "hands" of an atom, of "hands" of an atom,
 by which the atom by which the atom
 combines with other combines with other
 kinds of atoms. kinds of atoms.

 For example, For example,
 hydrogen and oxygen hydrogen and oxygen
 have one and two hands, have one and two hands,
 respectively, respectively,
 so that one atom so that one atom
 of oxygen combines of oxygen combines
 with two atoms of hydrogen with two atoms of hydrogen
 to form a molecule of water. to form a molecule of water.
 Accordingly the valence Accordingly the valence
 of hydrogen is 1, of hydrogen is 1,
 and that and that
 of oxygen is 2. of oxygen is 2.

 The atomic weights The atomic weights
 of hydrogen and oxygen are of hydrogen and oxygen are
 1 and 16, 1 and 16,
 respectively. respectively.
 The chemical The chemical
 equivalent of hydrogen equivalent of hydrogen
 is therefore is therefore
 1, and that 1, and that
 of oxygen is 8. of oxygen is 8.

|
 [The Elementary Charge [The Elementary Charge
 from the Viewpoint from the Viewpoint
 of Electrolysis] of Electrolysis]

 Suppose that, Suppose that,
 during an electrolysis, during an electrolysis,
 the mass of the mass of
 the separated element the separated element
 is M whose atomic weight is M whose atomic weight
 is A is A
 and its valence is v, and its valence is v,
 and the total electric charge and the total electric charge
 carried is Q. carried is Q.
 Since the mass M Since the mass M
 is proportional to the is proportional to the
 chemical equivalent A/v, chemical equivalent A/v,
 we have we have




 where 1/F is where 1/F is
 the proportionality constant. the proportionality constant.

 Now, let us consider Now, let us consider
 electrolysis electrolysis
 from the viewpoint from the viewpoint
 of atomism. of atomism.
 It is natural It is natural
 to consider that to consider that
 the charge carried the charge carried
 by an atom by an atom
 should be proportional should be proportional
 to the number to the number
 of "hand" of the atom, of "hand" of the atom,
 i.e. the valence. i.e. the valence.
 Let the quantity Let the quantity
 of charge carried of charge carried
 by one "hand" by one "hand"
 be a unit of charge, be a unit of charge,
 q. q.
 Therefore the total Therefore the total
 charge carried charge carried
 by an atom by an atom
 is vq. is vq.

 Let us think Let us think
 of the case of the case
 where electrolysis where electrolysis
 decomposes 1 mole decomposes 1 mole
 of element of element
 which includes which includes
 a number of atoms a number of atoms
 equal to Avogadro's constant equal to Avogadro's constant
 NA. NA.
 Thereby the total charge Thereby the total charge
 carried is carried is
 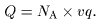
 Since Since
 M = A M = A
 in this case, in this case,
 we have we have


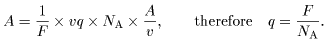

 from the above equation. from the above equation.

 The constant F The constant F
 is called is called
 Faraday's constant Faraday's constant
 whose value is measured whose value is measured
 to be to be


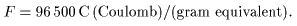

 This means This means
 that the electric charge that the electric charge
 necessary to separate necessary to separate
 1 gram equivalent 1 gram equivalent
 of any element of any element
 by electrolysis by electrolysis
 is 96500 C is 96500 C
 (Coulomb). (Coulomb).
 Here the gram equivalent Here the gram equivalent
 is the equivalent weight is the equivalent weight
 of an element of an element
 or a substance or a substance
 expressed in grams. expressed in grams.
 For hydrogen, For hydrogen,
 1 gram equivalent 1 gram equivalent
 is 1 gram of hydrogen. is 1 gram of hydrogen.
 In the case of In the case of
 the electrolysis the electrolysis
 of copper sulfate shown of copper sulfate shown
 in the in the
 above figure
above figure
 on this page, on this page,
 since the valence since the valence
 of copper is 2 of copper is 2
 and its atomic weight and its atomic weight
 is 63, is 63,
 1 gram equivalent 1 gram equivalent
 of copper is of copper is
 31.5 grams of elemental copper. 31.5 grams of elemental copper.

 From the above-mentioned From the above-mentioned
 result, we can see that result, we can see that
 the minimum unit the minimum unit
 of electric charge of electric charge
 in electrolysis in electrolysis
 is considered to be is considered to be




 This is just the This is just the
 elementary electric charge elementary electric charge
 or, or,
 more briefly, more briefly,
 the elementary charge. the elementary charge.

|
 [Millikan's Experiment]
[Millikan's Experiment]

 In 1909, In 1909,
 about 80 years about 80 years
 after the discovery of after the discovery of
 Faraday's law of electrolysis, Faraday's law of electrolysis,
 a precise measurement a precise measurement
 of elementary charge of elementary charge
 was carried out was carried out
 by R. A. Millikan by R. A. Millikan
 (USA, 1868 - 1953) (USA, 1868 - 1953)
 in a physical way. in a physical way.
 This is the famous This is the famous
 "Millikan oil "Millikan oil
 drop experiment". drop experiment".

 The schematic diagram The schematic diagram
 of the experimental set up of the experimental set up
 of Millikan's experiment of Millikan's experiment
 is shown in the following is shown in the following
 figure: figure:


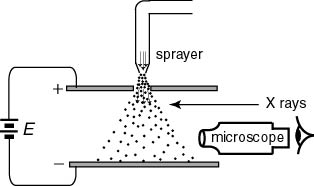

 Fine oil drops Fine oil drops
 are scattered are scattered
 between two metal plates between two metal plates
 with a sprayer. with a sprayer.
 The reason oil The reason oil
 was used instead water was used instead water
 was to avoid the quick was to avoid the quick
 evaporation of the drops. evaporation of the drops.
 The oil drops are The oil drops are
 usually charged usually charged
 during spraying, during spraying,
 but sometimes X-rays but sometimes X-rays
 are used are used
 to charge them sufficiently. to charge them sufficiently.

 Large oil drops Large oil drops
 which are unsuitable for measurement which are unsuitable for measurement
 fall quickly out fall quickly out
 of view of the microscope of view of the microscope
 and those of suitable and those of suitable
 sizes remain. sizes remain.

 Because there is Because there is
 the air between the plates, the air between the plates,
 the gravitation the gravitation
 and the viscosity and the viscosity
 resistance due to the air resistance due to the air
 work on an oil drop, work on an oil drop,
 so that it slowly falls down so that it slowly falls down
 at a constant at a constant
 speed. speed.
 This speed is measured This speed is measured
 through the microscope. through the microscope.
 If a voltage E If a voltage E
 applied between applied between
 the plates, the plates,
 an upward force acts an upward force acts
 on a charged drop on a charged drop
 and the speed of descent and the speed of descent
 changes. changes.
 The measurement The measurement
 of the speed of the speed
 is repeated many times, is repeated many times,
 varying varying
 the voltage E. the voltage E.

 Millikan measured Millikan measured
 the charges on the oil drops the charges on the oil drops
 in this experiment in this experiment
 and he found that they and he found that they
 are integral multiples are integral multiples
 of a minimum unit of a minimum unit
 of charge e of charge e
 whose value is whose value is
 agrees with that agrees with that
 of the elementary charge of the elementary charge
 obtained from the obtained from the
 experiment of electrolysis. experiment of electrolysis.

 Thus it became Thus it became
 clear that clear that
 the basic minimum element the basic minimum element
 of electricity of electricity
 is the elementary charge is the elementary charge
 e, e,
 whose currently whose currently
 accepted value is accepted value is


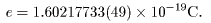

|
 Top
Top
|
|

 Go back to
the top page of Part 1. Go back to
the top page of Part 1.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|