
 Top of Part 2
Top of Part 2

 Last page
Last page

 Top of Part 3
Top of Part 3
  Top of Part 2
Top of Part 2
  Last page
Last page
  Top of Part 3
Top of Part 3
|
2-7: Summary of Part 2 |
 Let us summarize Let us summarize
 what we have what we have
 learned in Part 2. learned in Part 2.

|
 (1) (1) |
 Through the discoveries Through the discoveries
 of the radioactivity of the radioactivity
 and the decay and the decay
 of radioactive elements, of radioactive elements,
 it has been realized it has been realized
 that an atom that an atom
 has an internal structure has an internal structure
 and is not and is not
 eternally constant eternally constant
 but but
 changeable. changeable.

|
 (2) (2) |
 An atom has An atom has
 a structure in which a structure in which
 a massive nucleus a massive nucleus
 at the center at the center
 is surrounded by is surrounded by
 light electrons. light electrons.

|
 (3) (3) |
 The size of the atomic The size of the atomic
 nucleus is nucleus is
 very small; very small;
 its radius its radius
 is less than is less than
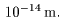

|
 (4) (4) |
 The atomic nucleus The atomic nucleus
 has also an has also an
 internal structure internal structure
 and is a changeable and is a changeable
 object. object.
 The proton The proton
 is one of the constituents is one of the constituents
 of the nucleus. of the nucleus.

|
 [The Difficulty in the Rutherford Model]
[The Difficulty in the Rutherford Model]

 In the solar system, In the solar system,
 the planets are the planets are
 revolving around revolving around
 the sun. the sun.
 In the Rutherford model, In the Rutherford model,
 the electrons are the electrons are
 considered considered
 to move around to move around
 the nucleus. the nucleus.

 The attractive force The attractive force
 between the sun between the sun
 and a planet is and a planet is
 the universal gravitation. the universal gravitation.
 On the other hand, On the other hand,
 the force through the force through
 which the nucleus which the nucleus
 attracts electrons attracts electrons
 is the Coulomb force. is the Coulomb force.
 One might naively One might naively
 suppose that suppose that
 this Coulomb attractive this Coulomb attractive
 force force
 and the centrifugal force and the centrifugal force
 due to the orbital motion due to the orbital motion
 of the electron of the electron
 are just in balance are just in balance
 to keep the system stable. to keep the system stable.
 However, However,
 this idea has this idea has
 a serious difficulty. a serious difficulty.

 Now, let us explain Now, let us explain
 this difficulty this difficulty
 taking the case of taking the case of
 hydrogen atom hydrogen atom
 in which an electron in which an electron
 (of mass m (of mass m
 and charge e ) and charge e )
 is moving around a proton. is moving around a proton.
 Suppose that, Suppose that,
 since the proton mass since the proton mass
 is large compared is large compared
 with that of electron, with that of electron,
 the proton stays the proton stays
 at rest at the origin at rest at the origin
 of the coordinate frame. of the coordinate frame.

|
 (A) (A)
 The balance The balance
 between the centrifugal between the centrifugal
 force and force and
 the Coulomb force the Coulomb force
 is written is written


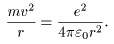
 The quantity The quantity
 on the left-hand side on the left-hand side
 is the strength of is the strength of
 the centrifugal force the centrifugal force
 and the right-hand side and the right-hand side
 the Coulomb force. the Coulomb force.
 Here Here
 r is the r is the
 radius of the orbit radius of the orbit
 of the electron, of the electron,
 i.e. the radius i.e. the radius
 of the hydrogen atom, of the hydrogen atom,
 and v is and v is
 the speed of the electron. the speed of the electron.
 The above equation The above equation
 is rewritten is rewritten


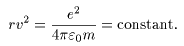
 Looking at this Looking at this
 equation only, equation only,
 we can think we can think
 that the radius that the radius
 of the hydrogen of the hydrogen
 is inversely proportional is inversely proportional
 to the square to the square
 of the speed of the speed
 of the electron, of the electron,
 so that the hydrogen so that the hydrogen
 atom can have atom can have
 an arbitrary radius an arbitrary radius
 depending depending
 on the speed v. on the speed v.
 However, the realistic However, the realistic
 hydrogen is not so; hydrogen is not so;
 the radius of the the radius of the
 realistic hydrogen realistic hydrogen
 atom looks atom looks
 constant always. constant always.

|
 (B) (B)
 According to According to
 the theory the theory
 of electromagnetism, of electromagnetism,
 any accelerating charged any accelerating charged
 particle radiates particle radiates
 electromagnetic waves, electromagnetic waves,
 with losing the energy. with losing the energy.
 Thus the electron Thus the electron
 in the Rutherford model in the Rutherford model
 must lose the energy must lose the energy
 to become out of balance to become out of balance
 and spiral inward, and spiral inward,
 and finally collapse and finally collapse
 on the proton. on the proton.
 This means This means
 that the hydrogen atom that the hydrogen atom
 in the Rutherford model in the Rutherford model
 must be unstable. must be unstable.
 In contrast to this, In contrast to this,
 however, a realistic however, a realistic
 hydrogen atom hydrogen atom
 is quite stable is quite stable
 and does not collapse. and does not collapse.

 Rutherford was well Rutherford was well
 aware of this difficulty, aware of this difficulty,
 but he but he
 chose to ignore chose to ignore
 it temporarily. it temporarily.
 Without solving Without solving
 this stability problem, this stability problem,
 we cannot look at we cannot look at
 the true feature the true feature
 of the microscopic world. of the microscopic world.

|
 Top
Top
|
|

 Go back to
the top page of Part 2. Go back to
the top page of Part 2.

 Go back to
the last page. Go back to
the last page.


 Go to
the top page of Part 3. Go to
the top page of Part 3.
|