
 Top of Part 3
Top of Part 3

 Next page
Next page
  Top of Part 3
Top of Part 3
  Next page
Next page
|
3-1: What is heat? |
 When an object When an object
 is heated, is heated,
 its temperature rises. its temperature rises.
 Everyone knows that, Everyone knows that,
 when an object when an object
 of higher temperature of higher temperature
 is in contact with is in contact with
 another of lower another of lower
 temperature, temperature,
 heat is transmitted heat is transmitted
 from the higher from the higher
 to the lower temperature to the lower temperature
 object. object.
 Here, what is heat? Here, what is heat?

 It is known well It is known well
 at present that at present that
 the heat transfer the heat transfer
 (heat conduction) is (heat conduction) is
 a transfer of energy; a transfer of energy;
 namely, heat namely, heat
 is a form of is a form of
 energy. energy.
 How can we explain this? How can we explain this?

 This problem This problem
 was just the clue was just the clue
 to make a relation to make a relation
 between the macroscopic between the macroscopic
 and the microscopic world. and the microscopic world.

|
 [The Phlogiston Theory, [The Phlogiston Theory,
 the Caloric Theory] the Caloric Theory]

 Up to the beginning Up to the beginning
 of the 19th century, of the 19th century,
 combustion combustion
 had been understood had been understood
 by supposing an invisible by supposing an invisible
 negative-mass negative-mass
 substance named substance named
 phlogiston. phlogiston.
 According to this theory, According to this theory,
 every combustible object every combustible object
 contains phlogiston contains phlogiston
 and combustion and combustion
 is explained as is explained as
 a process to lose a process to lose
 the phlogiston. the phlogiston.
 When such an object When such an object
 as mercury burns, as mercury burns,
 the mass would increase. the mass would increase.
 Looking at this fact, Looking at this fact,
 people thought people thought
 that the mass that the mass
 of the phlogiston of the phlogiston
 is negative. is negative.

 The heat conduction The heat conduction
 had been explained had been explained
 by introducing by introducing
 an imponderable fluid an imponderable fluid
 called caloric. called caloric.
 It was considered It was considered
 that that
 a higher temperature a higher temperature
 object contains object contains
 more caloric more caloric
 than a lower one, than a lower one,
 and the transfer and the transfer
 of the caloric implies of the caloric implies
 the heat conduction. the heat conduction.

 Lavoisier Lavoisier
 confirmed by confirmed by
 carrying out carrying out
 quantitative chemical quantitative chemical
 experiments precisely experiments precisely
 that combustion that combustion
 of an object of an object
 is a process is a process
 in which some in which some
 substances combine substances combine
 with others. with others.
 Thereby the theory Thereby the theory
 of phlogiston was denied. of phlogiston was denied.
 Moreover, looking at Moreover, looking at
 an ultimate supply an ultimate supply
 of heat of heat
 by friction, by friction,
 people suspected people suspected
 the caloric theory the caloric theory
 and abandoned it and abandoned it
 after Joule's experiment after Joule's experiment
 that will be that will be
 described below. described below.

|
 [Nature of Heat] [Nature of Heat]

 Using the apparatus Using the apparatus
 shown in the following shown in the following
 figure, figure,
 J. P. Joule J. P. Joule
 (UK, 1818 - 89) (UK, 1818 - 89)
 confirmed that confirmed that
 heat is heat is
 equivalent to equivalent to
 energy, energy,
 and measured and measured
 the mechanical equivalent the mechanical equivalent
 of heat of heat
 that represents that represents
 the quantitative relation the quantitative relation
 between heat between heat
 and mechanical energy. and mechanical energy.

|


|
The apparatus used
by Joule

 This is This is
 the apparatus used the apparatus used
 by Joule by Joule
 who confirmed who confirmed
 the equivalence between the equivalence between
 heat and heat and
 mechanical energy. mechanical energy.
 When the weights When the weights
 go down, go down,
 the impellers the impellers
 in a water tank in a water tank
 rotate to stir the water. rotate to stir the water.
 The potential energy The potential energy
 of the weights is of the weights is
 converted into converted into
 the kinematical energy the kinematical energy
 of the of the
 rotation of rotation of
 the impeller, the impeller,
 and the temperature and the temperature
 of the water rises. of the water rises.

|
 [The Mechanical Equivalent [The Mechanical Equivalent
 of Heat] of Heat]

 The unit to measure heat The unit to measure heat
 is cal (calorie). is cal (calorie).
 The heat required The heat required
 to raise the temperature to raise the temperature
 of 1 g of water by of 1 g of water by
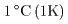

 from from

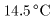
 to to

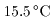

 at 1 atmospheric pressure at 1 atmospheric pressure
 is 1 cal. is 1 cal.
 The currently accepted value is The currently accepted value is


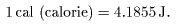

|
 [Statistical Mechanics] [Statistical Mechanics]

 As learned in As learned in
 Part 1: (Prologue: Atomic Nature
of Matter and Electricity,
Part 1: (Prologue: Atomic Nature
of Matter and Electricity,
 it became clear it became clear
 that that
 a matter consists a matter consists
 of a huge number of a huge number
 of molecules and atoms. of molecules and atoms.
 At first, At first,
 the motions of these the motions of these
 molecules and atoms are molecules and atoms are
 considered to be described considered to be described
 according to the laws of according to the laws of
 the classical theory, the classical theory,
 i.e. Newtonian mechanics and i.e. Newtonian mechanics and
 Maxwellian electromagnetism. Maxwellian electromagnetism.

 Although we cannot watch Although we cannot watch
 the motions of molecules the motions of molecules
 and atoms directly, and atoms directly,
 we can connect them we can connect them
 with the quantities with the quantities
 in the macroscopic world, in the macroscopic world,
 e.g. the pressure e.g. the pressure
 or the temperature, or the temperature,
 calculating an appropriate calculating an appropriate
 statistical average statistical average
 over a great number over a great number
 of degrees of of degrees of
 freedom of those molecules freedom of those molecules
 or atoms. or atoms.
 For example, For example,
 the pressure of a gas is the pressure of a gas is
 the average value the average value
 of the forces acting of the forces acting
 on the wall on the wall
 of the container of the container
 when the gas molecules when the gas molecules
 colliding with it. colliding with it.
 And the temperature And the temperature
 of the gas denotes of the gas denotes
 the average value the average value
 of the energies of of the energies of
 individual molecules. individual molecules.

 Thus the attempt Thus the attempt
 in which the various in which the various
 laws in the macroscopic world laws in the macroscopic world
 are derived are derived
 from the microscopic from the microscopic
 degrees of freedom degrees of freedom
 of individual molecules of individual molecules
 and atoms and atoms
 is just is just
 statistical mechanics statistical mechanics
 developed by developed by
 J. C. Maxwell J. C. Maxwell
 (UK, 1831 - 79) (UK, 1831 - 79)
 and and
 L. Boltzmann L. Boltzmann
 (Germany, 1844 - 1906). (Germany, 1844 - 1906).

|
 Top
Top
|
|
 Go back to
the top page of Part 3. Go back to
the top page of Part 3.

 Go to
the next page. Go to
the next page.
|