
 Top of Part 4
Top of Part 4

 Last page
Last page

 Next page
Next page
  Top of Part 4
Top of Part 4
  Last page
Last page
  Next page
Next page
|
4-2: The Atomic Spectra |

 The lights from discharge tubes The lights from discharge tubes
 or arcs across or arcs across
 various kinds of electrodes various kinds of electrodes
 with a high voltage potential with a high voltage potential
 being applied being applied
 represent line spectra represent line spectra
 which consist which consist
 of a numbers of bright sharp lines of a numbers of bright sharp lines
 characteristic of the gases characteristic of the gases
 involved in the tube involved in the tube
 or the matters of the electrodes. or the matters of the electrodes.
 Some examples are shown Some examples are shown
 in the pictures in the pictures
 below. below.

|

|
Examples of Atomic Spectra
 The sun light has The sun light has
 a continuous spectrum, a continuous spectrum,
 in which there are in which there are
 some absorption lines some absorption lines
 named the Fraunhofer lines named the Fraunhofer lines
 of which detailed explanations of which detailed explanations
 are omitted here. are omitted here.
 The spectra of hydrogen The spectra of hydrogen
 and mercury and mercury
 show some characteristic lines, show some characteristic lines,
 but the pictures here but the pictures here
 are not so good are not so good
 to look them clearly. to look them clearly.

|
 [Balmer's Formula] [Balmer's Formula]

 The group of lines The group of lines
 of the hydrogen spectrum of the hydrogen spectrum
 named the Balmer series named the Balmer series
 which are observed which are observed
 in the part from visible to in the part from visible to
 ultraviolet region ultraviolet region
 is shown in is shown in
 the following picture. the following picture.
 (Other series (Other series
 than the Balmer one, than the Balmer one,
 e.g. the Lymann, Paschen e.g. the Lymann, Paschen
 and Brackett series, and Brackett series,
 are found in the hydrogen are found in the hydrogen
 spectrum as well.) spectrum as well.)

|

|
 A line spectra of hydrogen atom
A line spectra of hydrogen atom
 (the Balmer series) (the Balmer series)

|
 A high school teacher, A high school teacher,
 J. J. Balmer J. J. Balmer
 (Switzerland, 1825 - 98) (Switzerland, 1825 - 98)
 discovered in 1885 that discovered in 1885 that
 the wavelengths of the spectral lines, the wavelengths of the spectral lines,
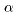 , ,
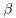 , ,
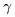 and
and
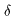 , ,

 in the Balmer series in the Balmer series
 shown shown
 in the above picture in the above picture
 are respectively written are respectively written


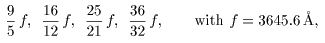

 and these are represented and these are represented
 by a simple formula by a simple formula


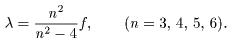

 This is called This is called
 Balmer's formula. Balmer's formula.
 It has been confirmed It has been confirmed
 that the other lines that the other lines
 in the Balmer series in the Balmer series
 than the above than the above
 , ,
 , ,
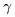
 and and
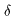
 also obey Balmer's also obey Balmer's
 formula mentioned above. formula mentioned above.

|
 [Rydberg's Formula] [Rydberg's Formula]

 In 1890, In 1890,
 Rydberg (Sweden) Rydberg (Sweden)
 found a very simple formula found a very simple formula
 which which
 can represent can represent
 the wavelengths the wavelengths
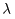 , ,
 not only in the not only in the
 hydrogen spectrum hydrogen spectrum
 but also in the spectra but also in the spectra
 of alkali atoms. of alkali atoms.
 This is This is
 Rydberg's formula Rydberg's formula


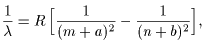

 where m where m
 and n are integers, and n are integers,
 and a and b and a and b
 are different are different
 for each series of spectra for each series of spectra
 but constant within a series. but constant within a series.
 The constant R The constant R
 is called the is called the
 Rydberg constant Rydberg constant
 whose value was obtained whose value was obtained
 from the experimental data from the experimental data
 for hydrogen as for hydrogen as


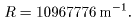

 Balmer's formula Balmer's formula
 is one of the special cases in is one of the special cases in
 Rydberg's formula. Rydberg's formula.
 Putting Putting
 a = b = 0 and a = b = 0 and
 m = 2 m = 2
 in Rydberg's formula, in Rydberg's formula,
 we can obtain we can obtain


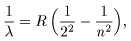

 which is equivalent which is equivalent
 to Balmer's formula. to Balmer's formula.

 We cannot explain these We cannot explain these
 Balmer's and Rydberg's Balmer's and Rydberg's
 formulae formulae
 with the classical theory. with the classical theory.
 At this point people At this point people
 necessitated necessitated
 making a big jump making a big jump
 to a new revolutionary theory. to a new revolutionary theory.

|
 Top
Top
|
|

 Go back to
the top page of Part 3. Go back to
the top page of Part 3.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|