
 Top of Part 3
Top of Part 3

 Next page
Next page
  Top of Part 3
Top of Part 3
  Next page
Next page
|
3-1: The Origin of the Nuclear Energy |
 [Exothermic [Exothermic
 Chemical Reactions] Chemical Reactions]

 Heat is evolved Heat is evolved
 in the chemical reaction in the chemical reaction
 in which hydrogen and oxygen in which hydrogen and oxygen
 are combined to be water; are combined to be water;
 i.e. the combustion i.e. the combustion
 of hydrogen. of hydrogen.
 Such chemical reaction Such chemical reaction
 in which heat is evolved in which heat is evolved
 is called exothermic is called exothermic
 reaction. reaction.
 The chemical equation The chemical equation
 of this reaction of this reaction
 for one mol of hydrogen for one mol of hydrogen
 is written is written


 Namely, when one mol of hydrogen Namely, when one mol of hydrogen
 burns, 286 kJ of heat is evolved. burns, 286 kJ of heat is evolved.

 Another example is Another example is


 where one mol of carbon where one mol of carbon
 is oxidated to be is oxidated to be
 carbon dioxide carbon dioxide
 with producing with producing
 394 kJ of heat. 394 kJ of heat.

 The above chemical equations, The above chemical equations,
 (1) and (2), are for (1) and (2), are for
 one mol of hydrogen and carbon, one mol of hydrogen and carbon,
 respectively. respectively.
 In order to compare these chemical In order to compare these chemical
 reactions with nuclear reactions, reactions with nuclear reactions,
 it is convenient to rewrite these it is convenient to rewrite these
 equations "for one molecule". equations "for one molecule".
 For this, let us divide For this, let us divide
 the heat production the heat production
 by the Avogadro constant by the Avogadro constant


 Then they are rewritten as Then they are rewritten as



 Equation (3) means that Equation (3) means that
 the process in which the process in which
 two hydrogen and one oxygen two hydrogen and one oxygen
 molecules combine to be molecules combine to be
 one water molecule one water molecule
 generates 3.0 eV energy emission. generates 3.0 eV energy emission.
 And Eq. (4) says that, And Eq. (4) says that,
 when a carbon atom combines with when a carbon atom combines with
 an oxygen molecule to be an oxygen molecule to be
 a carbon dioxiside molecule, a carbon dioxiside molecule,
 4.1 eV energy is released. 4.1 eV energy is released.

 As learned before, As learned before,
 eV is a unit of energy eV is a unit of energy
 extensively used extensively used
 in the atomic and nuclear world. in the atomic and nuclear world.
 It is the work done on an electron It is the work done on an electron
 that is accelerated through that is accelerated through
 a potential difference a potential difference
 of one volt. of one volt.
 Its value is Its value is


 Moreover, the units of energy, Moreover, the units of energy,
 keV and MeV, keV and MeV,
 are often used in the nuclear world; are often used in the nuclear world;
 the former is 1,000 times eV the former is 1,000 times eV
 and the latter 1,000,000 times eV. and the latter 1,000,000 times eV.

 We can understand that We can understand that
 "the energy evolved from "the energy evolved from
 one process of one process of
 an exothermic chemical reaction an exothermic chemical reaction
 is about 3 or 4 eV". is about 3 or 4 eV".

|
 [Exothermic [Exothermic
 Nuclear Reactions] Nuclear Reactions]

 Nuclei show Nuclei show
 various types of reaction: various types of reaction:
 For example, one nuclide For example, one nuclide
 splits splits
 into two or more fragments. into two or more fragments.
 This type of reaction is called This type of reaction is called
 nuclear fission. nuclear fission.
 Contrarily, two nuclides sometimes Contrarily, two nuclides sometimes
 combine with each other combine with each other
 to be a new nuclide. to be a new nuclide.
 This type of reaction is called This type of reaction is called
 nuclear fusion. nuclear fusion.
 There are many other types of There are many other types of
 reaction processes; reaction processes;
 they are generally called they are generally called
 nuclear reaction. nuclear reaction.

 Among these various types Among these various types
 of nuclear reactions, of nuclear reactions,
 there are some types of there are some types of
 exothermic reactions exothermic reactions
 which are sometimes called which are sometimes called
 "exoergic" reaction "exoergic" reaction
 in nuclear physics. in nuclear physics.

 The nucleus of deuterium The nucleus of deuterium
 atom is called deuteron atom is called deuteron
 which consists of which consists of
 a proton and a neutron. a proton and a neutron.
 It is represented by It is represented by
 a symbol "d". a symbol "d".
 The nuclear reaction The nuclear reaction
 in which two deuterons in which two deuterons
 bind with each other is bind with each other is
 an example of nuclear fusion. an example of nuclear fusion.
 This exoergic reaction is written as This exoergic reaction is written as

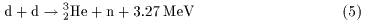
 If a neutron is If a neutron is
 absorbed in absorbed in
 the uranium-235 nucleus the uranium-235 nucleus
 (
(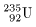
 ), ),
 it would split it would split
 into two fragments into two fragments
 of almost equal masses of almost equal masses
 and evolves some number and evolves some number
 of neutrons and of neutrons and
 energy Q. energy Q.
 One of the equations One of the equations
 for the processes is written for the processes is written

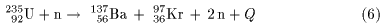
 This is an example of This is an example of
 nuclear fission. nuclear fission.

 Here it is quite interesting Here it is quite interesting
 how much the amount of the how much the amount of the
 energy emission Q energy emission Q
 is. is.
 It must be about 200 MeV. It must be about 200 MeV.
 Let us explain below why it is so. Let us explain below why it is so.
 The details of the mechanism The details of the mechanism
 of nuclear fission and fusion of nuclear fission and fusion
 will be given will be given
 on the following pages. on the following pages.

|
 [The Origin [The Origin
 of the Nuclear Energy] of the Nuclear Energy]

 Let us take up Let us take up
 the d-d fusion reaction the d-d fusion reaction
 shown by the above Eq. (5) shown by the above Eq. (5)
 as an example. as an example.
 Since the experimental value of Since the experimental value of
 the binding energy the binding energy
 of deuteron is 2.2246 MeV, of deuteron is 2.2246 MeV,
 the sum of the binding energies the sum of the binding energies
 of the two deuterons of the two deuterons
 before the reaction before the reaction
 (on the left-hand side of Eq.(5)) (on the left-hand side of Eq.(5))
 is 4.449 MeV. is 4.449 MeV.
 On the other hand, On the other hand,
 the experimental value of the experimental value of
 the binding energy of the binding energy of
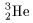
 is 7.719 MeV. is 7.719 MeV.
 Therefore the total Therefore the total
 binding energy after the reaction binding energy after the reaction
 (on the right-hand side of Eq. (5)) is (on the right-hand side of Eq. (5)) is
 3.27 MeV (= 7.719 - 4.449) 3.27 MeV (= 7.719 - 4.449)
 larger than the binding energy larger than the binding energy
 before the reaction before the reaction
 (on the left-hand side of Eq. (5)). (on the left-hand side of Eq. (5)).
 Thereby the total mass Thereby the total mass
 decreases after the reaction decreases after the reaction
 and the mass defect corresponding and the mass defect corresponding
 to the above increase of to the above increase of
 the binding energy the binding energy
 occurs. occurs.
 This mass defect is This mass defect is
 released as the heat (or energy) released as the heat (or energy)
 production in production in
 this exothermic (or exoergic) this exothermic (or exoergic)
 process. process.

 Next, we shall discuss Next, we shall discuss
 the change of the binding energy the change of the binding energy
 before and after the fission before and after the fission
 of uranium-235 of uranium-235
 (
(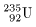
 ) )
 shown by Eq. (6). shown by Eq. (6).
 As seen in the figure on As seen in the figure on
 Page 2-4, [Comparison between Mass formula and Experiment] ,
Page 2-4, [Comparison between Mass formula and Experiment] ,
 the binding energy per nucleon the binding energy per nucleon
 in nuclei around in nuclei around
 A = 240 A = 240
 is about 7.5 MeV. is about 7.5 MeV.
 On the other hand, On the other hand,
 that that
 in nuclei around in nuclei around
 A = 120 A = 120
 is about 8.5 MeV. is about 8.5 MeV.
 Accordingly, if a uranium nucleus Accordingly, if a uranium nucleus
 splits into two fragments splits into two fragments
 with almost equal masses, with almost equal masses,
 the binding energy per nucleon the binding energy per nucleon
 would increase would increase
 by about 1 MeV by about 1 MeV
 and the total mass of and the total mass of
 the fission fragments the fission fragments
 would decrease would decrease
 by the corresponding amount. by the corresponding amount.
 This loss of mass This loss of mass
 (or mass defect) (or mass defect)
 is converted into is converted into
 the heat (or energy) product the heat (or energy) product
 Q. Q.
 Since an energy of about 1 MeV Since an energy of about 1 MeV
 per nucleon is released, per nucleon is released,
 the total energy the total energy
 Q Q
 would be more than would be more than
 200 MeV. 200 MeV.

 According to the above discussions, According to the above discussions,
 it becomes clear that it becomes clear that
 "the origin of nuclear energy "the origin of nuclear energy
 is the change of nuclear masses", is the change of nuclear masses",
 and it is based on and it is based on
 the principle of the principle of
 Einstein's Mass-Energy Equivalence.
Einstein's Mass-Energy Equivalence.

 If the total binding energy If the total binding energy
 after the reaction is larger than after the reaction is larger than
 before, the total sum of the masses before, the total sum of the masses
 of the reaction products of the reaction products
 becomes smaller than that before becomes smaller than that before
 the reaction. the reaction.
 This decrease in mass This decrease in mass
 is converted into an energy, is converted into an energy,
 so that the process would be so that the process would be
 an exothermic (exoergic) reaction. an exothermic (exoergic) reaction.

|
 [The Origin of the Heat [The Origin of the Heat
 in Exothermic Chemical Reaction, in Exothermic Chemical Reaction,
 Law of Energy Conservation] Law of Energy Conservation]

 We have learned that, We have learned that,
 if hydrogen and carbon burn if hydrogen and carbon burn
 in oxygen gas, heat or energy in oxygen gas, heat or energy
 is evolved. is evolved.
 What is the origin of this What is the origin of this
 heat or energy? heat or energy?
 The principle of the The principle of the
 heat production in chemical heat production in chemical
 reaction is reaction is
 just the same as just the same as
 that in the nuclear reaction. that in the nuclear reaction.

 The hydrogen molecule The hydrogen molecule
 is a bound system of two hydrogen atoms. is a bound system of two hydrogen atoms.
 The mass of a hydrogen molecule The mass of a hydrogen molecule
 is slightly smaller than is slightly smaller than
 the sum of the masses of two hydrogen atoms. the sum of the masses of two hydrogen atoms.
 Converting this Converting this
 difference (= mass defect) difference (= mass defect)
 into an energy with into an energy with
 Einstein's Mass-Energy Equivalence,
Einstein's Mass-Energy Equivalence,
 we have the binding energy we have the binding energy
 of the hydrogen molecule. of the hydrogen molecule.

 In the process of In the process of
 combustion of hydrogen combustion of hydrogen
 represented by Eq. (3), represented by Eq. (3),
 the total mass the total mass
 after the reaction after the reaction
 is slightly smaller is slightly smaller
 than before, than before,
 and this decrease in mass and this decrease in mass
 is transformed into the heat emission is transformed into the heat emission
 in the exothermic reaction. in the exothermic reaction.

 Strictly speaking, Strictly speaking,
 conservation conservation
 of mass does not hold of mass does not hold
 even in a chemical reaction. even in a chemical reaction.
 Both in chemical and Both in chemical and
 nuclear reactions, nuclear reactions,
 the energy of the total system the energy of the total system
 with converting mass into energy with converting mass into energy
 conserves before and after conserves before and after
 the reaction. the reaction.

|
 [Huge Amount of Nuclear Energy] [Huge Amount of Nuclear Energy]

 Comparing Eq. (3) with (5), Comparing Eq. (3) with (5),
 and Eq. (4) with (6), and Eq. (4) with (6),
 we can easily understand we can easily understand
 that the nuclear reactions that the nuclear reactions
 yield a huge amount of energy yield a huge amount of energy
 in comparison with in comparison with
 ordinary combustion processes. ordinary combustion processes.

 As explained above, As explained above,
 "the energy evolved from "the energy evolved from
 an exothermic chemical reaction an exothermic chemical reaction
 like combustion of hydrogen or carbon like combustion of hydrogen or carbon
 is about 3 or 4 eV". is about 3 or 4 eV".
 In contrast, In contrast,
 d-d fusion reaction d-d fusion reaction
 shown by Eq. (5) releases shown by Eq. (5) releases
 3.27 MeV of energy. 3.27 MeV of energy.
 It is about several million times It is about several million times
 as large as the ordinary combustion. as large as the ordinary combustion.

 In the fission of In the fission of
 uranium-235 shown by Eq. (6), uranium-235 shown by Eq. (6),
 the more energy than 200 MeV the more energy than 200 MeV
 is emitted. is emitted.
 It is about several 100 million times It is about several 100 million times
 as large as an ordinary as large as an ordinary
 chemical reaction. chemical reaction.

 Thus, the nuclear energy Thus, the nuclear energy
 released in nuclear fission and fusion released in nuclear fission and fusion
 is several million is several million
 or or
 100 million 100 million
 times as large as times as large as
 an ordinary chemical an ordinary chemical
 reaction like a combustion reaction like a combustion
 process. process.

|
 Top
Top
|
|
 Go back to
the top page of Part 3 Go back to
the top page of Part 3

 Go to
the next page. Go to
the next page.
|