
 Top of Part 3
Top of Part 3

 Last page
Last page

 Next page
Next page
  Top of Part 3
Top of Part 3
  Last page
Last page
  Next page
Next page
|
3-4: Nuclear Fusion |
 On the top page On the top page
 of the present Part 3, of the present Part 3,
 3-1:The Origin of the Nuclear Energy,
3-1:The Origin of the Nuclear Energy,
 we took we took
 an example an example
 of exothermic (exoergic) of exothermic (exoergic)
 nuclear reaction nuclear reaction
 in which two deuterons in which two deuterons
 combine together combine together
 to form helium-3
( to form helium-3
( ) )
 and emit a neutron (n) and emit a neutron (n)
 and release an energy of 3.27 MeV. and release an energy of 3.27 MeV.
 This fusion reaction process This fusion reaction process
 is written as is written as

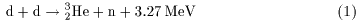
 We interpreted We interpreted
 the reason why such a big amount the reason why such a big amount
 of energy as 3.27 MeV of energy as 3.27 MeV
 is released in this process is released in this process
 as follows: as follows:
 After this fusion reaction, After this fusion reaction,
 the binding energy of the produced the binding energy of the produced
 helium-3
( helium-3
( ) )
 is 7.719 MeV is 7.719 MeV
 which is considerably larger which is considerably larger
 than the sum of than the sum of
 the binding energies the binding energies
 of the two deuterons of the two deuterons
 before the reaction, before the reaction,
 4.449 MeV. 4.449 MeV.

 Like this, Like this,
 the binding energy the binding energy
 as a function of the mass number as a function of the mass number
 A A
 fluctuates much fluctuates much
 in the region of light nuclei. in the region of light nuclei.
 Utilizing this fluctuation, Utilizing this fluctuation,
 one can get large amounts of one can get large amounts of
 energy through energy through
 nuclear fusion reaction. nuclear fusion reaction.

 Let us see Let us see
 the experimental data of the experimental data of
 the binding energies the binding energies
 of the light nuclei of the light nuclei
 in detail in detail
 in the following in the following
 figure. figure.

|

|
 [Nuclear Binding Energies] [Nuclear Binding Energies]

 This figure shows This figure shows
 the experimental data the experimental data
 of the binding energies of the binding energies
 per nucleon per nucleon
 in the region of light nuclei. in the region of light nuclei.
 This is essentially the same This is essentially the same
 as the last one as the last one
 at the bottom of the page:
"2-3: Mass of Nuclei, Binding Energy", at the bottom of the page:
"2-3: Mass of Nuclei, Binding Energy",
 but the data for nuclei but the data for nuclei
 of smaller mass number of smaller mass number
 than 20 than 20
 can be seen in further detail. can be seen in further detail.

|
 Casting a glance at the Casting a glance at the
 above figure, above figure,
 we can see that, if we can see that, if
 very light nuclides, very light nuclides,
 e.g. e.g.
 proton (p), deuteron (d), proton (p), deuteron (d),
 helium-3 ( helium-3 (
 ) )
 or triton (t), or triton (t),
 combine together to form combine together to form
 such a slightly heavier one such a slightly heavier one
 as as
 helium-4 ( helium-4 (

 ), ),
 a large amount of energy a large amount of energy
 would be released. would be released.

 (Needless to say, (Needless to say,
 proton, deuteron, proton, deuteron,
 helium-3, triton, and helium-3, triton, and
 helium-4 are helium-4 are
 the atomic nuclei of the atomic nuclei of
 hydrogen(protium), deuterium, hydrogen(protium), deuterium,
 helium-3, tritium helium-3, tritium
 and helium-4 atoms, respectively.) and helium-4 atoms, respectively.)

 In addition to In addition to
 the above d-d reaction (Eq. (1)), the above d-d reaction (Eq. (1)),
 the following reactions the following reactions



 are also assumed to be capable are also assumed to be capable
 to be utilized for fusion power. to be utilized for fusion power.
 Among these reactions, Among these reactions,
 the easiest to utilize the easiest to utilize
 is the d-t reaction is the d-t reaction
 (Eq. (2)). (Eq. (2)).
 (See the following (See the following
 figure.) figure.)

|

|
 [The d-t Reaction] [The d-t Reaction]

 If deuteron and triton If deuteron and triton
 collide at a high speed, collide at a high speed,
 a fusion reaction occurs a fusion reaction occurs
 and a large amount of and a large amount of
 energy (= 17.58 MeV) energy (= 17.58 MeV)
 is released. is released.

|
 The device The device
 in which nuclear fusion reaction in which nuclear fusion reaction
 takes place continuously takes place continuously
 to extract energy to extract energy
 is called a is called a
 nuclear fusion reactor. nuclear fusion reactor.
 Fusion is clearly a more Fusion is clearly a more
 "mass-energy efficient" process "mass-energy efficient" process
 than fission, than fission,
 because the difference of because the difference of
 the binding energies per nucleon the binding energies per nucleon
 before and after the reaction before and after the reaction
 is larger in fusion than in fission. is larger in fusion than in fission.
 In this sense, fusion In this sense, fusion
 is more favorable than fission. is more favorable than fission.
 While experiments for While experiments for
 utilization of utilization of
 fusion reaction continue, fusion reaction continue,
 no actual fusion reactor no actual fusion reactor
 exist yet. exist yet.

 To develope To develope
 a reactor for practical use, a reactor for practical use,
 many difficult problems many difficult problems
 should be overcome. should be overcome.
 Among them, Among them,
 the most important the most important
 is the confinement of is the confinement of
 plasma. plasma.

|
 [Confinement of Plasma] [Confinement of Plasma]

 Let us take up Let us take up
 the above d-t reaction the above d-t reaction
 as an example. as an example.
 d and t are d and t are
 the nuclei of the nuclei of
 deuterium and deuterium and
 tritium elements, tritium elements,
 respectively. respectively.
 In these elements, In these elements,
 the nuclei are surrounded the nuclei are surrounded
 by electrons by electrons
 and usually exist and usually exist
 as electrically neutral. as electrically neutral.
 Namely, d and t are ions Namely, d and t are ions
 of these neutral elements. of these neutral elements.
 The gases of these ions The gases of these ions
 are called plasma. are called plasma.

 Therefore both d and t Therefore both d and t
 in the plasma have in the plasma have
 +e +e
 charges, charges,
 and the Coulomb repulsive force and the Coulomb repulsive force
 works between them. works between them.
 In order for d and t In order for d and t
 to fuse together, to fuse together,
 they must get closer to each other they must get closer to each other
 and collide at a very high speed and collide at a very high speed
 overcoming the Coulomb overcoming the Coulomb
 repulsive force. repulsive force.
 For this, For this,
 one must confine the plasma one must confine the plasma
 at a state of at a state of
 extremely high temperature extremely high temperature
 and pressure; and pressure;
 e.g. higher than 0.1 GK (= giga Kelvin). e.g. higher than 0.1 GK (= giga Kelvin).

 How can we confine How can we confine
 such an enormously high temperature such an enormously high temperature
 and pressure of plasma? and pressure of plasma?
 This is none other than This is none other than
 the problem of the problem of
 plasma confinement. plasma confinement.
 To carry out a Research and Development To carry out a Research and Development
 of a fusion power reactor, of a fusion power reactor,
 the construction of the construction of
 ITER ITER
 (International Thermonuclear (International Thermonuclear
 Experimental Reactor) Experimental Reactor)
 has internationally been negotiated. has internationally been negotiated.

|
 [Resources for Fusion Power] [Resources for Fusion Power]

 Deuterium Deuterium
 is one of naturally occurring is one of naturally occurring
 isotopes of hydrogen isotopes of hydrogen
 whose abundance is whose abundance is
 0.015% (in number of atoms). 0.015% (in number of atoms).
 It can be taken It can be taken
 from heavy water, from heavy water,
 whoes resources are whoes resources are
 quite rich on the earth. quite rich on the earth.
 It is easy to separate deuterium It is easy to separate deuterium
 from hydrogen isotopes. from hydrogen isotopes.

 In contrast to this, In contrast to this,
 tritium is not naturally tritium is not naturally
 occurring. occurring.
 It must be produced It must be produced
 in an artificial way; in an artificial way;
 for this purpose, for this purpose,
 the fusion reaction the fusion reaction
 shown by shown by
 the above Eq. (3) the above Eq. (3)
 considered to be suitable. considered to be suitable.
 The resources of lithium The resources of lithium
 (Li) appears (Li) appears
 rather rich on the earth. rather rich on the earth.

|
 Top
Top
|
|

 Go back to
the top page of Part 3. Go back to
the top page of Part 3.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|