
 Top of Part 3
Top of Part 3

 Last page
Last page

 Next page
Next page
  Top of Part 3
Top of Part 3
  Last page
Last page
  Next page
Next page
|
3-3: Nuclear Fission |
 On the previous page:
3-1:The Origin of the Nuclear Energy, On the previous page:
3-1:The Origin of the Nuclear Energy,
 the fission of uranium-235 the fission of uranium-235
 ( (
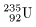 ) )
 was taken up as an example was taken up as an example
 of nuclear exothermic reaction, of nuclear exothermic reaction,
 in which a huge amount of energy in which a huge amount of energy
 is released when neutrons is released when neutrons
 being bomberded against uranium-235 being bomberded against uranium-235
 nuclei to split them into nuclei to split them into
 several fragments. several fragments.
 One of the equations for these fission One of the equations for these fission
 processes is written as processes is written as

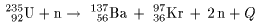
 Here Here
 Q Q
 is the released energy is the released energy
 which is more than which is more than
 200 MeV per one process. 200 MeV per one process.
 "2n" on the right-hand side "2n" on the right-hand side
 means that two neutrons means that two neutrons
 are simultaneously emitted. are simultaneously emitted.
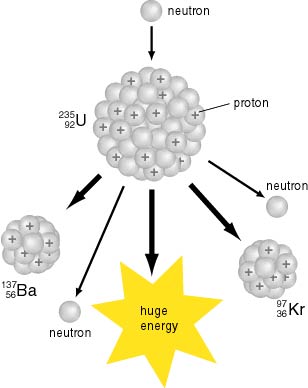
 A schematic sketch A schematic sketch
 of this fission process of this fission process
 is drawn as the following figure. is drawn as the following figure.

|
|
 [Schematic Drawing of the Fission [Schematic Drawing of the Fission
 Process of Uranium-235] Process of Uranium-235]

 When neutrons are bombarded When neutrons are bombarded
 against uanium-235, against uanium-235,
 the uranium-235 nucleus absorbs the uranium-235 nucleus absorbs
 a neutron to be a uranium-236 a neutron to be a uranium-236
 and it and it
 splits into splits into
 two fragments of almost equal two fragments of almost equal
 masses masses
 with emitting with emitting
 some neutrons simultaneously some neutrons simultaneously
 and evolving and evolving
 a huge amount of energy a huge amount of energy
 larger than 200 MeV. larger than 200 MeV.

 This is nothing but This is nothing but
 one of the examples one of the examples
 of fission of uranium-235. of fission of uranium-235.
 It does not always It does not always
 split into Ba and Kr split into Ba and Kr
 but usually into two fragments but usually into two fragments
 with almost equal masses. with almost equal masses.
 The number of emitted neutrons The number of emitted neutrons
 is also not always constant is also not always constant
 but it distributes over one but it distributes over one
 to several. to several.
 Then the emitted energy is Then the emitted energy is
 not always constant not always constant
 but is almost 200 MeV. but is almost 200 MeV.

|
 The above equation is The above equation is
 one of the examples one of the examples
 of fission of uranium-235. of fission of uranium-235.
 There are many other processes There are many other processes
 of which examples are of which examples are
 as follows: as follows:

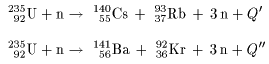

|
 [The Discovery of Nuclear Fission] [The Discovery of Nuclear Fission]

 Rutherford Rutherford
 carried out alpha-particle carried out alpha-particle
 scattering experiments scattering experiments
 by bombarding the alpha rays by bombarding the alpha rays
 emerging from radioactive Polonium (Po) emerging from radioactive Polonium (Po)
 or Radium (Ra) elements or Radium (Ra) elements
 onto various nuclei. onto various nuclei.
 Thereby he succeeded Thereby he succeeded
 in the first
artificial transmutation of elements (1919). in the first
artificial transmutation of elements (1919).

 Since then, experiments Since then, experiments
 of alpha-particle scattering of alpha-particle scattering
 on nuclei on nuclei
 had been repeated extensively. had been repeated extensively.
 Chadwick investigated Chadwick investigated
 the mysterious penetrating radiation the mysterious penetrating radiation
 emitted from a beryllium (Be) target emitted from a beryllium (Be) target
 which is bombarded by which is bombarded by
 high-energy alpha particles high-energy alpha particles
 from a Po source. from a Po source.
 Finally he concluded Finally he concluded
 that this mysterious radiation is that this mysterious radiation is
 a neutral particle a neutral particle
 with protonic mass. with protonic mass.
 This was
the discovery of neutron This was
the discovery of neutron
 (1932). (1932).

 O. Hahn O. Hahn
 (Germany, 1879 - 1968) and (Germany, 1879 - 1968) and
 F. Strassmann F. Strassmann
 (Germany, 1902 - 68) (Germany, 1902 - 68)
 carried out the experiments carried out the experiments
 to bombard neutrons to bombard neutrons
 from (Po-Be) source from (Po-Be) source
 against uranium. against uranium.
 They observed an important They observed an important
 phenomenon in which phenomenon in which
 the uranium nucleus the uranium nucleus
 bombarded by neutrons bombarded by neutrons
 splits into two large splits into two large
 fragments. fragments.
 Among the reaction products, Among the reaction products,
 they found they found
 Barium (Z = 56) Barium (Z = 56)
 and Lanthanum and Lanthanum
 (Z = 57). (Z = 57).

 L. Meitner L. Meitner
 (Germany, 1878 - 1968), (Germany, 1878 - 1968),
 a former collaborator of Hahn, a former collaborator of Hahn,
 and her nephew and her nephew
 O. R. Frisch, O. R. Frisch,
 who had fled to Sweden who had fled to Sweden
 from Nazi Germany, from Nazi Germany,
 were reported these experimental results were reported these experimental results
 by a letter. by a letter.
 They analyzed these results They analyzed these results
 and immediately recognized and immediately recognized
 the significance. the significance.
 They were convinced that They were convinced that
 the experimental results the experimental results
 are due to are due to
 the neutron-induced the neutron-induced
 nuclear fission nuclear fission
 and they published this idea. and they published this idea.

 This was the first This was the first
 discovery of nuclear fission. discovery of nuclear fission.

|
 [The Mechanism of Nuclear Fission] [The Mechanism of Nuclear Fission]

 As explained on the page: As explained on the page:

 3-1: The Origin of the Nuclear Energy,
3-1: The Origin of the Nuclear Energy,
 if such a heavy nucleus if such a heavy nucleus
 as uranium-235 as uranium-235
 splits into two fragments splits into two fragments
 of almost equal mass, of almost equal mass,
 each of them would be bound more each of them would be bound more
 tightly than the original nucleus. tightly than the original nucleus.
 In that case, an energy excess In that case, an energy excess
 is brought about is brought about
 and the state after the reaction and the state after the reaction
 becomes more favorable becomes more favorable
 than before the reaction. than before the reaction.
 Accordingly, Accordingly,
 a heavy nucleus has a heavy nucleus has
 always a possibility of always a possibility of
 nuclear fission. nuclear fission.

 Using the idea of Using the idea of
 Liquid Drop Model,
Liquid Drop Model,
 let us explain let us explain
 in what mechanism in what mechanism
 such a heavy nucleus such a heavy nucleus
 as uranium-235 fission. as uranium-235 fission.

 The nucleus is The nucleus is
 a many-nucleon system a many-nucleon system
 consisting of protons and neutrons. consisting of protons and neutrons.
 It can be assumed to be It can be assumed to be
 something like something like
 a drop of water a drop of water
 or a raindrop. or a raindrop.
 A heavy nucleus A heavy nucleus
 is therefore considered to be is therefore considered to be
 very "soft" and "flabby" very "soft" and "flabby"
 and easy to deform. and easy to deform.
 However, since a strong nuclear force However, since a strong nuclear force
 works between nucleons works between nucleons
 and it plays a role of and it plays a role of
 surface tension, surface tension,
 the ground state of a nucleus the ground state of a nucleus
 is rather stable is rather stable
 and does not directly fission. and does not directly fission.
 If some amount of energy is given If some amount of energy is given
 to a nucleus to be to a nucleus to be
 in an exited state, in an exited state,
 then it easily deforms then it easily deforms
 and fissions. and fissions.
 You would be able to understand You would be able to understand
 the fissioning mechanism the fissioning mechanism
 by the by the
 following two figures. following two figures.

|

|
 [Fission Mechanism by Liquid Drop Model] [Fission Mechanism by Liquid Drop Model]

 If such a heavy nucleus If such a heavy nucleus
 as uranium-235 as uranium-235
 absorbs a neutron, absorbs a neutron,
 it is in an excited state. it is in an excited state.
 The soft nucleus is easy The soft nucleus is easy
 to deform. to deform.
 Sometimes it becomes Sometimes it becomes
 dumbbells shape dumbbells shape
 and splits into and splits into
 two droplets. two droplets.
 At that time At that time
 the energy excess the energy excess
 is released. is released.

|

|
 [Schematic Graph of Energy in Nuclear Fission]
[Schematic Graph of Energy in Nuclear Fission]

 This diagram schematically shows This diagram schematically shows
 the potential energy for nuclear fission the potential energy for nuclear fission
 as a function of as a function of
 the degree of nuclear deformation the degree of nuclear deformation
 or the average separation or the average separation
 of the two fission fragments. of the two fission fragments.
 The ground state of the nucleus The ground state of the nucleus
 is confined within the valley is confined within the valley
 of potential barrier of potential barrier
 and is stable. and is stable.
 When it is excited When it is excited
 to be close to to be close to
 the top of the barrier, the top of the barrier,
 it penetrates the barrier it penetrates the barrier
 with the
tunnel effect with the
tunnel effect
 and run down along the potential and run down along the potential
 slope to fission. slope to fission.

|
 [The Chain Reaction] [The Chain Reaction]

 As shown in the equation at As shown in the equation at
 the top of this page, the top of this page,
 the fission of a uranium-235 the fission of a uranium-235
 is accompanied by the emission is accompanied by the emission
 of neutrons. of neutrons.
 There are several types of There are several types of
 fission processes fission processes
 in the uranium-235 fission, in the uranium-235 fission,
 in each of which one, two or three in each of which one, two or three
 neutrons are emitted; neutrons are emitted;
 on average, 2.5 neutrons on average, 2.5 neutrons
 are released. are released.
 These neutrons are capable to These neutrons are capable to
 cause further fission cause further fission
 of uranium-235. of uranium-235.
 If each fission causes If each fission causes
 an average of more an average of more
 than one further fission, than one further fission,
 an avalanche of fission reactions an avalanche of fission reactions
 will result. will result.
 This is called This is called
 the chain reaction. the chain reaction.

 As soon as Hahn and Strassmann As soon as Hahn and Strassmann
 discovered nuclear fission, discovered nuclear fission,
 many physicists many physicists
 noticed the possibility noticed the possibility
 of the chain reaction. of the chain reaction.
 If the chain reaction If the chain reaction
 occurs very rapidly occurs very rapidly
 in an uncontrolled way, in an uncontrolled way,
 a violent explosion follows. a violent explosion follows.
 Then it can be used for a military Then it can be used for a military
 purpose; i.e. an atomic purpose; i.e. an atomic
 or strictly speaking a nuclear or strictly speaking a nuclear
 bomb. bomb.
 If the chain reaction If the chain reaction
 is well controlled, is well controlled,
 then it can be used then it can be used
 as a power source. as a power source.

 The first experiment The first experiment
 of nuclear chain reaction of nuclear chain reaction
 was carried out was carried out
 in 1942 in 1942
 by E. Fermi by E. Fermi
 (Italy, USA, 1901 - 54), (Italy, USA, 1901 - 54),
 who had fled to US who had fled to US
 from Fascists Italy, from Fascists Italy,
 and his colleagues. and his colleagues.

|
 [Nuclear Reactor] [Nuclear Reactor]

 A device in which controlled nuclear A device in which controlled nuclear
 fission takes place fission takes place
 to produce heat or energy to produce heat or energy
 is called a nuclear reactor. is called a nuclear reactor.
 It is the main facility It is the main facility
 of a nuclear power station. of a nuclear power station.
 The first nuclear reactor The first nuclear reactor
 was constructed was constructed
 by Fermi by Fermi
 and coworkers and coworkers
 at the University of Chicago at the University of Chicago
 in 1942. in 1942.

|
 [Other Kinds of Fissionable Materials] [Other Kinds of Fissionable Materials]

 An elemental isotope that undergoes An elemental isotope that undergoes
 induced fission induced fission
 when being struck by a free neutron when being struck by a free neutron
 is usually called "fissionable". is usually called "fissionable".
 Of course, the most popular Of course, the most popular
 fissionable material fissionable material
 is uranium-235 is uranium-235
 (
(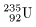 ). ).

 One of the other One of the other
 well-known fissionable well-known fissionable
 materials is materials is
 pultonium-239 pultonium-239
 (
(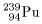 ), ),
 which is usually produced which is usually produced
 in a reactor in a reactor
 from uranium-238. from uranium-238.
 (Uranium-238 is not (Uranium-238 is not
 fissionable, so that fissionable, so that
 it does not "burn" it does not "burn"
 in a reactor.) in a reactor.)
 While almost all pultonium While almost all pultonium
 is artificially produced, is artificially produced,
 extremely tiny amount extremely tiny amount
 is found naturally. is found naturally.

 Another kind of Another kind of
 element element
 that can be used that can be used
 as a nuclear fuel as a nuclear fuel
 in reactors in reactors
 is thorium, is thorium,
 which occurs naturally. which occurs naturally.

|
 Top
Top
|
|

 Go back to
the top page of Part 3. Go back to
the top page of Part 3.

 Go back to
the last page. Go back to
the last page.

 Go to
the next page. Go to
the next page.
|